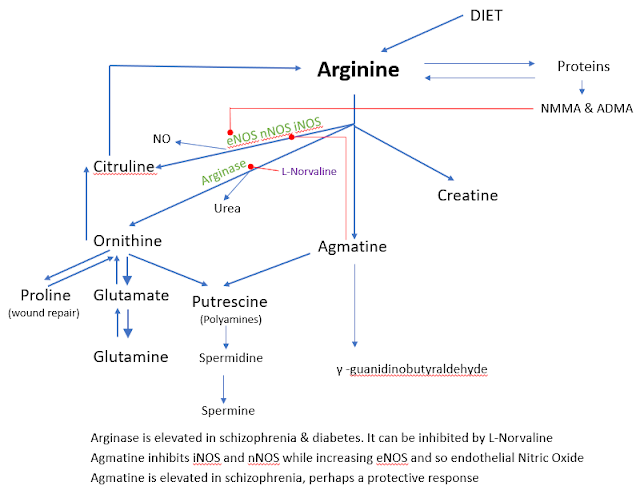
Today’s post again shows that many issues raised in previous posts keep on coming back – is that good news? Only you can decide.
I
start with the “old chestnut” (English idiom to imply “a tired old story”) of
the Autism Tsunami.
Then
we see what has come up in the world of autism interventions in the research in
the last 3 weeks, most of which regular readers will already be aware of.
·
Autism Tsunami –
real or not?
·
Vitamin D
·
Bumetanide
·
Ibudilast
·
Niclosamide
·
Non-invasive
brain stimulation
· Simvastatin
I noted the research about autism incidence coming from Northern Ireland because it was published in the Belfast News Letter. These days it has a tiny subscription, but I am one of those who know it is the world's oldest English-language general daily newspaper still in publication, having first been printed in 1737. In 1972 a bomb warning was called in to the paper's office and, as people evacuated, an explosion went off nearby killing several people and injuring many more. Back in the early 1990s, when some people in Northern Ireland were still blowing up others with bombs, I made a visit to Northern Ireland to meet the management of this newspaper.
Their recent
article on autism incidence is very well researched considering how only about
8,000 copies are published. Keep up the good work!
Idea
that 5% of all Northern Ireland's children are autistic is 'a fantasy' claims
international expert
Professor
Laurent Mottron was speaking to the News Letter following a claim that the rate
of autism in Northern Ireland is double the rate in the rest of the UK.
Back
in 2019 Prof Mottron had authored a report warning about a tsunami of
over-diagnosis, saying that soon "the definition of autism may get too
vague to be meaningful, trivializing the condition"
“If this trend holds, the objective difference
between people with autism and the general population will disappear in less
than 10 years," he had said then – and has now indicated that this
“fuzziness” is what’s helping swell the numbers in Northern Ireland.
Meanwhile Jill
Escher, the president of the National Council on Severe Autism, takes a different
view.
She says that
evidence indicates the "skyrocketing" rate of autism in Northern
Ireland is real, adding: "It boggles my mind that it is not the subject of
the highest possible alarm and inquiry."
"One
in 20 children in Northern Ireland of school age has a diagnosis of
autism," he told MPs.
"[It
is] one in 57 in the rest of the UK. The need in Northern Ireland is
significantly different."
To
put that in perspective, that would mean 5% of Northern Irish children are
diagnosed with autism, compared with 1.8% in the rest of the UK.
Prof Mottron, a psychiatrist based at Montreal University, told the News Letter "numbers such as 5% are pure fantasy... these numbers correspond to the part of the general population which has less overt socialisation, which has minimally to do with prototypical autism".
There is a "current fuzziness of autism diagnosis and over-inclusivity," he said, leading to "a situation of perfect confusion between autistic traits and prototypical autism" (that is, mixing up people who exhibit some tendencies of autistic people with people who actually have the full-blown condition).
"The scientific 'quasi consensus'
would be around 1% everywhere on the planet,” he added.
So on one side we have Jill Escher and
her NCSA and on the other we have a French/Canadian researcher. This time Laurent Mottron but in my blog posts I quoted Éric Fombonne.
A paper
that was mentioned both in my blog and critiqued by Jill about autism incidence
and cost just got retracted. In reality
a better word is “cancelled.” The 3
authors are very much in the politically incorrect camp of the autism debate.
I was surprised it ever got published.
Controversial ‘cost of autism’ paper retracted
Citing methodological issues and undeclared conflicts of
interest, an autism journal has retracted a paper that forecast the prevalence
and cost of autism.
The retraction note, posted last week, comes two years after Spectrum reported on backlash surrounding the paper, which was published in the Journal of Autism and Developmental Disorders in July 2021. A month after publication, the journal added an editor’s note that the study was under investigation because of criticisms of its conclusions.
“I am glad to see that it was retracted, although at a pace
that maybe is a bit frustrating in terms of how long it took. But it was the
right choice,” says Brittany Hand, associate professor of health and
rehabilitation sciences at Ohio State University in Columbus.
Outside experts who reviewed the paper on the journal’s
behalf found that it misrepresented the rise in autism diagnoses and gave
“insufficient attention” to some potential causes of the increase, such as
improved surveillance and changes to the diagnostic criteria. The authors also
used “higher estimates and assumptions that inflated costs,” according to the
retraction note.
The authors — Mark Blaxill, Toby Rogers and Cynthia Nevison —
all disagree with the journal’s decision, the note also says.
The cancelled paper is here:-
Autism Tsunami: the Impact of Rising Prevalence on the Societal Cost of Autism in the United States
I assume
Blaxill was the driving force behind all the math, because he is the ex-
management consultant, with a son with severe autism that his dad attributes to
vaccines.
What I found
bizarre in their paper was that they has a prevention scenario, based on what
they think has already happened in rich parts of California, where they think
autism incidence is falling. It is not
falling, all that is happening is that wealthy Californians are paying for
treatment using insurance or their own money, and no longer burdening the
State.
The
“rainbow” researchers that wanted the paper retracted think that preventing
autism is akin to eugenics and Dr Mengele. According to Peter, treating autism
is good, while Dr Josef Mengele, byname Todesengel (German: “Angel of Death”)
was as bad as you can get.
Jill Escher
and her NCSA think that you cannot prevent autism. According to Peter, you can both minimize the
incidence and severity of autism.
A bugbear of our reader Tanya is that the NCSA have a pet hate of facilitated communication and in particular the rapid prompting method (RPM). This method worked for Tanya’s son and it opened the door to independent, un-facilitated communication.
Always keep
an open mind.
“our Prevention scenario is based on real rates observed
among wealthy white and Asian children in the California DDS. Severe ASD prevalence has flattened and even
declined among these children since birth year 2000, suggesting that wealthy
parents have been making changes that effectively lower their children’s risk
of developing ASD. The Prevention scenario assumes that these parental
strategies and opportunities already used by wealthy parents to lower their
children’s risk of ASD can be identified and made available rapidly to lower
income children and ethnic minorities, who are currently experiencing the most
rapid growth in ASD prevalence”
New Paper Makes Case that Autism Tsunami May Threaten American Economy
A major weakness in the analysis was the “Prevention
Scenario” in which future costs were projected based on “what might be possible
if strategies for reducing ASD risk are identified and addressed in the near
future.” As I think everyone
knows, at this time there is no way to prevent autism. But the authors
use the observation that autism in the DDS is declining among wealthier white
families, and thus “suggesting that wealthy parents have been making changes
that effectively lower their children’s risk of developing ASD.” No, it’s far more likely that
wealthier families are not entering their children into the system because they
access services through insurance and school districts instead.
Vitamin D as
a cause of autism has been discussed for decades. As the title below puts it – a never-ending story. Our reader Seth Bittker even wrote a paper about it. He later wrote a paper about the use Acetaminophen/Paracetamol in children under two as a risk factor in developing autism. Good work Seth!
Maternal Vitamin D deficiency and brain functions: a never-ending story
A large number of
observational studies highlighted the prevalence rates of vitamin D
insufficiency and deficiency in many populations as pregnant women. Vitamin D
is well known to have a crucial role in differentiation and proliferation, as
well as neurotrophic and neuroprotective actions in brain. Then, this
micronutrient can modulate the neurotransmission and synaptic plasticity. Recent results from animal and
epidemiological studies indicated that maternal vitamin D deficiency is
associated with a wide range of neurobiological disease including autism,
schizophrenia, depression, multiple sclerosis or developmental defect.
The aim of this review is to provide a state of the art on the effect of
maternal vitamin D deficiency on brain functions and development.
4.2.2. Autism
Autism spectrum disorder (ASD) is
a complex neurodevelopmental disease with repetitive behaviour and difficulties
in social interaction, communication and learning. Several murine studies and
cohorts have demonstrated that early exposure to low levels of VD during
pregnancy could be a risk factor for ASD. In 2019, Ali et al. aimed to find out
the impact of a maternal VDD on early postnatal, adolescent and adult
offspring. By assessing righting reflex and negative geotaxis, they found out
that the pups from deficient dams showed a delay in their motor development.
P12 rats from deficient females also exhibited increased ultrasound
vocalization indicating an alteration in their vocal communication. Adolescent
and young adult rats displayed an altered stereotyped repetitive behaviour as
they had a reduced digging behaviour. Adolescent rats had less social
interaction with longer latency to interact, which was not found in adult rats;
however, adults were more hyperactive but showed no anxiety like
behaviour. In another animal study,
maternal VDD induced an increase in the vocalizations of the pups accompanied
with a decrease in cortical FoxP2, decrease in social behaviour and impaired
learning and memory were observed in adult males (Table 1). Using data from the
Stockholm youth cohort, Magnusson et al. examined a population of 4-17-year-old
children exposed to low levels of VD during gestation and was able to report a
positive association between maternal VDD and ASD. Analysing the same cohort,
Lee et al. suggested that high levels of VD during pregnancy were associated
with a moderate decrease in risk of ASD in the offspring. A prospective study
of a multi-ethnic cohort in the Netherlands (generation R study) has also shown
an association between maternal mid-gestation VDD and a two-fold increase in
the risk of autism in children (Table 2). Interestingly, VD supplementation seems to clinically
improve ASD symptoms of affected children.
People do
associate this blog with Bumetanide. Yet
another paper has been published showing the benefits of this therapy for
autism.
Highlights
·
We investigated the nonlinear brain connectivity and
topological changes in brain networks of people with autism spectrum disorders
(ASD) after a three-month course of bumetanide treatment.
·
We found statistically significant differences
between pre and post intervention in the connectivity patterns using repeated
measures analysis of variance (ANOVA).
·
We found that the number of strong connections in
response to sad image stimuli seem to be less compared with that of the other
two stimuli, especially in the central area.
·
We found that the changes in brain connectivity
between pre and post intervention is more significant in response to sad image
stimuli.
Emerging evidence suggests that cognitive impairment associated with brain network disorders in people with autism could be improved with medications such as bumetanide. However, the extent to which bumetanide is effective in improving brain function in these individuals has not been adequately studied. The main purpose of this study is to investigate the nonlinear brain connectivity and topological changes in brain networks of people with autism spectrum disorders (ASD) after a three-month course of bumetanide treatment. We used electroencephalography (EEG) data of nine participants recorded during the face emotion recognition activity in two stages before and after bumetanide treatment. Brain connectivity matrix was calculated using a neural network-based estimator. Graph criteria and statistical tests have been used to determine the effects of bumetanide treatment on children and adolescents with autism. Bumetanide treatment significantly alters the brain connectivity networks based on stimuli type. Differences in brain connectivity related to the sad stimuli are more significant. The most of the significant changes of the strength graph metric was in the occipital electrodes and electrodes related to the right hemisphere. These results suggest that bumetanide may affect effective connectivity and be used a promising treatment for improving social interactions in patients with autism. It also suggests that brain connectivity patterns can be considered as a neural marker to be used in the development of new therapies.
I have also covered in
sometimes painful details the potential to treat autism and increase cognitive
function using PDE (Phosphodiesterase) inhibitors. One of our psychiatrist
readers is a huge fan of Pentoxifylline and takes it himself.
I was recently asked how
to obtain Ibudilast. It is approved in
Japan as an asthma drug. Sometimes it is called Ketas and you can get it from
an “International Pharmacy” in Germany/Switzerland if you have a
prescription.
I also wrote about
repurposing Roflumilast, which as Daxas is approved all over the world as a therapy for severe asthma (COPD). This drug at a 1/5th dose has been
patented as a cognitive enhancer.
Autism spectrum disorder (ASD) is categorized as a
neurodevelopmental disorder, presenting with a variety of aetiological and
phenotypical features. Ibudilast is known to produce beneficial effects in
several neurological disorders including neuropathic pain, multiple sclerosis,
etc. by displaying its neuroprotective and
anti-inflammatory properties. Here, in our study, the pharmacological outcome
of ibudilast administration was investigated in the prenatal valproic acid
(VPA)-model of ASD in Wistar rats.
Methods
Autistic-like symptoms were induced in Wistar male pups of dams
administered with Valproic acid (VPA) on embryonic day 12.5. VPA-exposed male
pups were administered with two doses of ibudilast (5 and10 mg/kg) and all the
groups were evaluated for behavioral parameters like social interaction,
spatial memory/learning, anxiety, locomotor activity, and nociceptive
threshold. Further, the possible neuroprotective effect of ibudilast was
evaluated by assessing oxidative stress, neuroinflammation (IL-1β, TNF-α, IL-6,
IL-10) in the hippocampus, % area of Glial fibrillary acidic protein
(GFAP)-positive cells and neuronal damage in the cerebellum.
Key findings: Treatment with ibudilast significantly
attenuated prenatal VPA exposure associated social interaction and spatial
learning/memory deficits, anxiety, hyperactivity, and increased nociceptive
threshold, and it decreased oxidative stress markers, pro-inflammatory markers
(IL-1β, TNF-α, IL-6), and % area of GFAP-positive cells and restored neuronal
damage.
Conclusions
Ibudilast treatment has restored crucial ASD-related behavioural
abnormalities, potentially through neuroprotection. Therefore, benefits of
ibudilast administration in animal models of ASD suggest that ibudilast may have therapeutic
potential in the treatment of ASD.
I have also written widely about repurposing certain
anti-parasite medicines to treat autism. This is not because I think parasites
cause autism, it is the secondary modes of action.
Autism
Spectrum Disorders (ASD) are a complex set of neurodevelopmental manifestations
which present in the form of social and communication deficits. Affecting a
growing proportion of children worldwide, the exact pathogenesis of this
disorder is not very well understood, and multiple signaling pathways have been
implicated. Among them, the ERK/MAPK pathway is critical in a number of
cellular processes, and the normal functioning of neuronal cells also depends
on this cascade. As such, recent studies have increasingly focused on the
impact this pathway has on the development of autistic symptoms. Improper ERK
signaling is suspected to be involved in neurotoxicity, and the same might be
implicated in autism spectrum disorders (ASD), through a variety of effects
including mitochondrial dysfunction and oxidative stress. Niclosamide, an antihelminthic
and anti-inflammatory agent, has shown potential in inhibiting this pathway,
and countering the effects shown by its overactivity in inflammation.
While it has previously been evaluated in other neurological disorders like
Alzheimer’s Disease and Parkinson’s Disease, as well as various cancers by
targeting ERK/MAPK, it’s efficacy in autism has not yet been evaluated. In this
article, we attempt to discuss the potential role of the ERK/MAPK pathway in
the pathogenesis of ASD, specifically through mitochondrial damage, before
moving to the therapeutic potential of niclosamide in the disorder, mediated by
the inhibition of this pathway and its detrimental effects of neuronal
development.
Note
that in earlier posts I explored RASopathies as potentially treatable types of
intellectual disability (ID). We also have RAS-dependent cancers as a discrete
treatable sub-type of cancer.
The ERK/MAPK pathway is known to
interact with multiple genes that have been implicated in autism, and genome-wide association analysis of the
same have supported these findings. As such, a dysregulation of this pathway has been found to result
in many CNS disorders, including ASD-related syndromes, in many studies.
These syndromes are
collectively known as Rasopathies, due to the fact that the affected genes
include those encoding for elements which function together with Ras, a
G-protein responsible for activating ERKs (Levitt and Campbell 2009; Tidyman
and Rauen 2009). It has been found that ASD is linked to the occurrence of many
Rasopathies, and there have been multiple reports suggesting the possible
relation of ERK/MAPK pathway defects with the incidence of ASD (Vithayathil et
al. 2018; Aluko et al. 2021).
Moreover, a detailed study has found that single nucleotide polymorphisms
(SNPs) in the ERK/MAPK-related genes are more common in subjects presenting
with idiopathic ASD.
Niclosamide is an FDA-approved
antihelminthic drug which is routinely used to treat tapeworm infections by inhibiting their mitochondrial oxidative
phosphorylation and ATP production. In addition, it has long been known to have
significant immunomodulating activity, and has been shown to inhibit a number
of signaling pathways, including the Wingless-related integration site
(Wnt)/β-catenin, nuclear factor kappa B (Nf-κB), signal transducer and
activator of transcription 3 (STAT3), and mammalian target of rapamycin (mTOR)
(Chen et al. 2018). However, while these targets are known to be rather
well-characterized in terms of the effect that niclosamide has on them, there
are also other targets, including the phosphoinositode 3 kinase/Akt (PI3K/Akt)
and ERK/MAPK pathways, that are seen to be downregulated by the agent. Hence, given the possible relation of
the ERK pathway in autism, there has been interest in the potential role of
niclosamide in the management of the prognosis of ASD. This article aims
to discuss the possible therapeutic benefit of niclosamide in the treatment of
autism spectrum disorders.
Now I know that parents like the idea of treating autism with various gadgets you can strap on to your head – things like Transcranial Magnetic Stimulation (TMS). I must say I liked my old post on Photobiomodulation/cold laser/low level laser therapy.
Epiphany: Low Level Laser Therapy
(LLLT) for Autism – seems to work in Havana
From China we have a new round-up
paper, but the full text does not yet seem to be ready.
Non-invasive brain stimulation for Patient
with Autism: A Systematic Review and Meta-Analysis
Objective: To
comprehensively evaluate the efficacy of non-invasive brain stimulation (NIBS)
in patients with autism spectrum disorder (ASD) in randomized controlled trials
(RCT),providing reference for future research on the same topic.
Methods:Five
databases were searched (Pubmed,Web of science,Medline,Embase and Cochrane
library) and track relevant references,Meta-analysis was performed using RevMan
5.3 software.
Results: Twenty-two
references(829
participants) were included. The results of meta analysis showed that, NIBS had
positive effects on repetitive and stereotypical behaviors, cognitive function
and executive function in autistic patients. Most of the included
studies had a moderate to high risk of bias, Mainly because of the lack of
blinding of subjects and assessors to treatment assignment, as well as the lack
of continuous observation of treatment effects.
Conclusions:
Available evidence
supports an improvement in some aspects of NIBS in patients with ASD. However,
due to the quality of the original studies and significant publication bias,
these evidences must be treated with caution. Further large multicenter
randomized double-blind controlled trials and appropriate follow-up
observations are needed to further evaluate the specific efficacy of NIBS in
patients with ASD.
Unfortunately, the Chinese have
concluded that most of these studies are not reliable. So no laser for me to go
out and buy just yet.
No need to dent your bank balance with the next therapy. We are back to one of the world's most prescribed and therefore affordable drugs, its Simvastatin (Zocor).
There is masses of information in this blog about the potential to treat sub-types of autism with Atorvastatin, Simvastatin or Lovastatin. They are each slightly different.
Purpose: To study the effect of simvastatin on behavioral performance in a rat model of autism, and its effect on hippocampal brain-derived BDNF-TrkB pathway.
Methods: Twelve rats with valproic acid (VPA)-induced autism were randomly divided into model group and simvastatin group, while six healthy rats served as normal control group. Rats in the simvastatin group received the drug (5 mg/kg) via i.p. route, while rats in model group and normal control group were injected with equivalent volume of normal saline in place of simvastatin. Capacity for interaction and repetitive stereotyped behavior, as well as results of Morris water maze test were determined for each group. The expressions of BDNF-TrkB proteins were assayed with immunoblotting.
Results: The frequencies of sniffing normal saline, alcohol and rat urine were significantly higher in model and simvastatin rats than in normal rats, but they were significantly lower in simvastatin-treated rats than in model rats (p < 0.05). There was higher duration of turning, jumping and grooming in the model group and simvastatin group than in the normal rats, but the duration was significantly reduced in simvastatin rats, relative to model rats. Escape latency times was significantly longer in model and simvastatin rats than in controls, but number of target quadrant crossings was significantly reduced. However, escape latency time was lower in simvastatin rats than in model rats, but number of target quadrant crossings was significantly higher. The model and simvastatin rats had down-regulated levels of BDNF and TrkB protein, relative to control rats, but there were markedly higher levels of these proteins in simvastatin-treated rats than in model rats.
Conclusion: Simvastatin improves the
behavioral performance of autistic rats by regulating BDNF/TrkB signal axis.
This finding may be useful in the development of new drugs for treating autism.
Conclusion
What is the conclusion? Well, I could
say give up reading the new research and just read my old posts. It seems you are not going to miss very much.
Of course, back in the real world, it is true that things do take time to change and after a few decades the leap might be taken from the research to the doctor’s office.
There already is plenty of research on the causes of autism and what steps can be taken by those who want to treat aspects of it. It is far from a complete picture, but it is enough to get started. There are no guarantees of success, but if you want 100% certainty you will wait forever.