Shutting the stable door after the horse has bolted
Today’s post is a summary of what I found interesting in the latest research. Many items have been touched on previously.
The topic of maternal treatment to prevent future
autism did come up in some recent comments on this blog. Two of the recent
papers cover this very subject. One uses agmatine, from my autism PolyPill
therapy, while the other used choline.
Auditory sound sensitivity is a complex subject and
today we see the potential role impaired myelination and Nav1.2 ion channels
can play.
A Chinese study reconfirms the elevated level of androgen hormones in autism.
Apigenin which was covered in an earlier post is
shown to help “autistic” mice in the popular BTBR model. This is a model where
the corpus callosum is entirely absent.
Self-injury is a recuring nightmare for many with
severe autism and today we look at a possible correlation with dopamine
transporter binding abnormalities.
We start with easier subject matter and leave the
hard parts for later in the post.
Preventing future autism
It may seem like too late to be talking about
preventing autism, but it is a recurring subject. Today we have two new ideas that have
appeared in the literature, and both are very simple. One is choline and other
agmatine; both are used in the treatment of already existing autism.
Maternal
choline to prevent autism
“maternal choline supplementation may be sufficient to blunt some of the behavioral and neurobiological impacts of inflammatory exposures in utero, indicating that it may be a cheap, safe, and effective intervention for neurodevelopmental disorders.”
Maternal infection has
long been described as a risk factor for neurodevelopmental disorders,
especially autism spectrum disorders (ASD) and schizophrenia. Although many pathogens do not cross
the placenta and infect the developing fetus directly, the maternal immune
response to them is sufficient to alter fetal neurodevelopment, a phenomenon
termed maternal immune activation (MIA). Low maternal choline is also a risk factor for
neurodevelopmental disorders, and most pregnant people do not receive enough of
it. In addition to its role in neurodevelopment, choline is capable of
inducing anti-inflammatory signaling through a nicotinic pathway. Therefore, it
was hypothesized that maternal choline supplementation would blunt the
neurodevelopmental impact of MIA in offspring through long- term instigation of
cholinergic anti-inflammatory signaling.
To model MIA in rats, the viral mimetic
polyinosinic:polycytidylic acid (poly(I:C)) was used to elicit a maternal
antiviral innate immune response in dams both with and without choline
supplementation. Offspring were reared to both early and late adolescent stages
(postnatal days 28 and 50, respectively), where cognition and anxiety-related
behaviors were examined. After behavioral testing, animals were euthanized, and
their prefrontal cortices (PFCs) were collected for analysis. MIA offspring
demonstrated sex-specific patterns of altered cognition and repetitive
behaviors, which were modulated by maternal choline supplementation. Choline supplementation also
bolstered anti-inflammatory signaling in the PFCs of MIA animals at both early
and late adolescent stages. These findings suggest that maternal choline
supplementation may be sufficient to blunt some of the behavioral and
neurobiological impacts of inflammatory exposures in utero, indicating that it
may be a cheap, safe, and effective intervention for neurodevelopmental
disorders.
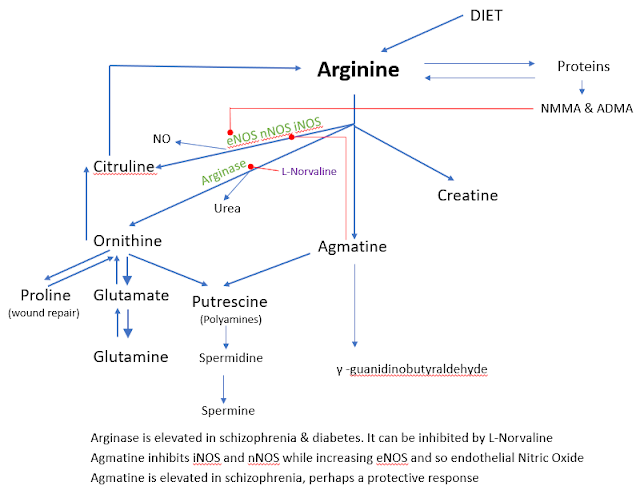
Prenatal Agmatine to prevent autism
Agmatine is a cheap bodybuilder supplement also
used in psychiatry that has been extensively covered in this blog. Here we see
how in a popular mouse model it can prevent autism.
Background: According to reports, prenatal
exposure to valproic acid can induce autism spectrum disorder (ASD)-like
symptoms in both humans and rodents. However, the exact cause and therapeutic
method of ASD is not fully understood. Agmatine (AGM) is known for its
neuroprotective effects, and this study aims to explore whether giving agmatine
hydrochloride before birth can prevent autism-like behaviors in mouse offspring
exposed prenatally to valproic acid.
Methods: In this study, we investigated
the effects of AGM prenatally on valproate (VPA)-exposed mice. We established a
mouse model of ASD by prenatally administering VPA. From birth to weaning, we
evaluated mouse behavior using the marble burying test, open-field test, and
three-chamber social interaction test on male offspring.
Results: The results showed prenatal use
of AGM relieved anxiety and hyperactivity behaviors as well as ameliorated
sociability of VPA-exposed mice in the marble burying test, open-field test,
and three-chamber social interaction test, and this protective effect might be
attributed to the activation of the ERK/CREB/BDNF signaling pathway.
Conclusion: Therefore, AGM can effectively reduce the likelihood of offspring
developing autism to a certain extent when exposed to VPA during pregnancy,
serving as a potential therapeutic drug.
This builds on an earlier paper that
first identified the benefit.
Agmatine rescues autistic
behaviors in the valproic acid-induced animal model of autism
Highlights
• Single
treatment of agmatine rescues social impairment in the VPA-induced animal model
of autism.
• Effect
of agmatine in social improvement in the VPA model is induced from agmatine
itself, not its metabolite.
• Agmatine
rescues repetitive and hyperactive behavior, and seizure susceptibility in the
VPA model.
• Overly
activated ERK1/2 in the brain of the VPA model is relieved by agmatine.
Apigenin
50mg of Apigenin
1g of dried parsley
15-20g of dried chamomile flowers
I have
previously written about Apigenin, which is an OTC supplement. There has been
another paper recently published about it. There is a logical connection with the maternal choline therapy from above.
What does
Apigenin have in common with Choline? α7-nAChRs
Choline is
interesting because it acts as both a precursor for acetylcholine synthesis and
it is a neuromodulator itself.
Choline is
activates α7-nAChRs, alpha-7 nicotinic acetylcholine receptors.
These
receptors are extremely important in learning and sensory processing. They also play a key role in inflammation and
signaling via the vagus nerve.
Apigenin is
a flavonoid found in many plants, fruits, and vegetables. It has been shown to
have a number of health benefits, including anti-inflammatory and antioxidant
effects. Apigenin has also been shown to interact with α7-nAChRs.
Studies have
shown that apigenin can:
Enhance α7-nAChR function: Apigenin has been shown to
increase the activity of α7-nAChRs. This may be due to its ability to bind to a
specific site on the receptor.
Protect α7-nAChRs from damage: Apigenin may also help to
protect α7-nAChRs from damage caused by oxidative stress.
Studying the involvement of nicotinic acetylcholine receptors
(nAChRs), specifically α7-nAChRs, in neuropsychiatric brain disorders such as
autism spectrum disorder (ASD) has gained a growing interest. The flavonoid apigenin (APG) has been
confirmed in its pharmacological action as a positive allosteric modulator of
α7-nAChRs. However, there is no research describing the pharmacological
potential of APG in ASD. The aim of this study was to evaluate the effects of
the subchronic systemic treatment of APG (10–30 mg/kg) on ASD-like repetitive
and compulsive-like behaviors and oxidative stress status in the hippocampus
and cerebellum in BTBR mice, utilizing the reference drug aripiprazole (ARP, 1
mg/kg, i.p.). BTBR mice pretreated with APG (20 mg/kg) or ARP (1 mg/g, i.p.)
displayed significant improvements in the marble-burying test (MBT),
cotton-shredding test (CST), and self-grooming test (SGT) (all p < 0.05).
However, a lower dose of APG (10 mg/kg, i.p.) failed to modulate behaviors in
the MBT or SGT, but significantly attenuated the increased shredding behaviors
in the CST of tested mice. Moreover,
APG (10–30 mg/kg, i.p.) and ARP (1 mg/kg) moderated the disturbed levels of
oxidative stress by mitigating the levels of catalase (CAT) and superoxide
dismutase (SOD) in the hippocampus and cerebellum of treated BTBR mice.
In patch clamp studies in hippocampal slices, the potency of choline (a
selective agonist of α7-nAChRs) in activating fast inward currents was
significantly potentiated following incubation with APG. Moreover, APG markedly
potentiated the choline-induced enhancement of spontaneous inhibitory
postsynaptic currents. The
observed results propose the potential therapeutic use of APG in the management
of ASD. However, further preclinical investigations in additional models
and different rodent species are still needed to confirm the potential
relevance of the therapeutic use of APG in ASD.
Altered acidity (pH) levels inside the brain
I found it intriguing that a large study has examined the altered acidity (pH) levels inside the brain of those with neurological disorders.
For all the disorders other than autism there was a
clear pattern of low pH, which means increased acidity.
For autism certain autism models exhibited
decreased pH and increased lactate levels, but others showed the opposite
pattern, reflecting subpopulations within autism.
Altered brain energy metabolism is an acknowledged feature
of autism, so we should not be surprised to find altered levels of acidity.
The easy reading version:
Brain Acidity Linked With Multiple
Neurological Disorders
The study itself:
Increased levels of lactate, an end-product of glycolysis, have been proposed as a potential surrogate marker for metabolic changes during neuronal excitation. These changes in lactate levels can result in decreased brain pH, which has been implicated in patients with various neuropsychiatric disorders. We previously demonstrated that such alterations are commonly observed in five mouse models of schizophrenia, bipolar disorder, and autism, suggesting a shared endophenotype among these disorders rather than mere artifacts due to medications or agonal state. However, there is still limited research on this phenomenon in animal models, leaving its generality across other disease animal models uncertain. Moreover, the association between changes in brain lactate levels and specific behavioral abnormalities remains unclear. To address these gaps, the International Brain pH Project Consortium investigated brain pH and lactate levels in 109 strains/conditions of 2,294 animals with genetic and other experimental manipulations relevant to neuropsychiatric disorders. Systematic analysis revealed that decreased brain pH and increased lactate levels were common features observed in multiple models of depression, epilepsy, Alzheimer’s disease, and some additional schizophrenia models. While certain autism models also exhibited decreased pH and increased lactate levels, others showed the opposite pattern, potentially reflecting subpopulations within the autism spectrum. Furthermore, utilizing large-scale behavioral test battery, a multivariate cross-validated prediction analysis demonstrated that poor working memory performance was predominantly associated with increased brain lactate levels. Importantly, this association was confirmed in an independent cohort of animal models. Collectively, these findings suggest that altered brain pH and lactate levels, which could be attributed to dysregulated excitation/inhibition balance, may serve as transdiagnostic endophenotypes of debilitating neuropsychiatric disorders characterized by cognitive impairment, irrespective of their beneficial or detrimental nature.
In conclusion, the
present study demonstrated that altered brain pH and lactate levels are
commonly observed in animal models of SZ, BD, ID, ASD, AD, and other
neuropsychiatric disorders. These findings provide further evidence supporting
the hypothesis that altered brain pH and lactate levels are not mere artifacts,
such as those resulting from medication confounding, but are rather involved in
the underlying pathophysiology of some patients with neuropsychiatric
disorders. Altered
brain energy metabolism or neural hyper- or hypoactivity leading to abnormal
lactate levels and pH may serve as a potential therapeutic targets for
neuropsychiatric disorders
Why would the brain be acidic (reduced pH)?
To function optimally mitochondria need adequate
oxygen and glucose. When performance is impaired, for example due to the lack
of Complex 1, mitochondria switch from OXPHOS (oxidative phosphorylation) to
fermentation to produce energy (ATP). Lactic acid is the byproduct and this
will lower pH.
Does brain pH matter?
It does matter and is linked to cognitive
impairments, headaches, seizures etc.
Many enzymes in the brain rely on a specific pH range to
function properly. Deviations from the ideal pH can hinder their activity,
impacting various neurochemical processes essential for brain function.
Some ion channels are pH sensitive.
Chemical buffers in the brain aim to regulate pH in the brain
· Carbonic Acid/Bicarbonate Buffer System: Similar to the blood, the brain utilizes this system to regulate pH.
· Organic Phosphates: These molecules, like creatine phosphate, can act as buffers in the brain by binding or releasing hydrogen ions.
These buffering systems work together to maintain a tightly controlled
pH range in both the blood (around 7.35-7.45) and the brain (slightly more
acidic than blood, around 7.0-7.3). Even slight deviations from this ideal
range can have significant consequences for cellular function.
Androgen Levels in Autism
Androgens are male hormones like testosterone, DHEA
and DHT, but females have them too, just at lower levels.
Drugs that reduce the level of these hormones are
called antiandrogens.
Finasteride reduces DHT and is used to treat hair
loss in men as Propecia. This drug was trialed in women, but failed to show a
benefit over the placebo.
The main use of Finasteride is for the treatment of
benign prostatic hyperplasia (BPH) in older men.
Women sometimes take antiandrogens like
Spironolactone to control acne.
Numerous studies have show elevated levels of males
hormones in both males and females with autism.
A recent paper was published on this very subject:
Androgen levels in autism spectrum disorders:
A systematic review and meta-analysis
Background:
Accumulating
evidence suggests that the autism spectrum disorder (ASD) population exhibits
altered hormone levels, including androgens. However, studies on the regulation of androgens,
such as testosterone and dehydroepiandrosterone (DHEA), in relation to sex
differences in individuals with ASD are limited and inconsistent. We conducted
the systematic review with meta-analysis to quantitatively summarise the blood,
urine, or saliva androgen data between individuals with ASD and controls.
Methods:
A systematic search was conducted for eligible
studies published before 16 January 2023 in six international and two Chinese
databases. We computed summary statistics with a random-effects model.
Publication bias was assessed using funnel plots and heterogeneity using I 2
statistics. Subgroup analysis was performed by age, sex, sample source, and
measurement method to explain the heterogeneity.
Results:
17 case-control studies (individuals with ASD,
825; controls, 669) were assessed. Androgen levels were significantly higher in individuals with ASD than
that in controls (SMD: 0.27, 95% CI: 0.06-0.48, P=0.01). Subgroup
analysis showed significantly elevated levels of urinary total testosterone,
urinary DHEA, and free testosterone in individuals with ASD. DHEA level was
also significantly elevated in males with ASD. Androgen levels, especially free testosterone, may be
elevated in individuals with ASD and DHEA levels may be specifically elevated
in males.
By coincidence I was just sent the paper below, showing the benefit of Finasteride in one model of autism.
Therapeutic effect of finasteride through its
antiandrogenic and antioxidant role in a propionic acid-induced autism model:
Demonstrated by behavioral tests, histological findings and MR spectroscopy
I do recall I think it was Tyler, long ago, writing
a comment about the potential to use Finasteride in autism.
Some very expensive antiandrogens have been used in
autism and this became rather controversial.
We saw in earlier posts that RORα/RORalpha/RORA is
a key mechanism where the balance between male and female hormones controls
some key autism gene.
The schematic illustrates a mechanism through which the observed reduction in RORA in autistic brain may lead to increased testosterone levels through downregulation of aromatase. Through AR, testosterone negatively modulates RORA, whereas estrogen upregulates RORA through ER.
Cerebellum and neurodevelopmental disorders:
RORα is a unifying force
Errors of cerebellar development are increasingly acknowledged as risk factors for neuro-developmental disorders (NDDs), such as attention deficit hyperactivity disorder (ADHD), autism spectrum disorder (ASD), and schizophrenia. Evidence has been assembled from cerebellar abnormalities in autistic patients, as well as a range of genetic mutations identified in human patients that affect the cerebellar circuit, particularly Purkinje cells, and are associated with deficits of motor function, learning and social behavior; traits that are commonly associated with autism and schizophrenia. However, NDDs, such as ASD and schizophrenia, also include systemic abnormalities, e.g., chronic inflammation, abnormal circadian rhythms etc., which cannot be explained by lesions that only affect the cerebellum. Here we bring together phenotypic, circuit and structural evidence supporting the contribution of cerebellar dysfunction in NDDs and propose that the transcription factor Retinoid-related Orphan Receptor alpha (RORα) provides the missing link underlying both cerebellar and systemic abnormalities observed in NDDs. We present the role of RORα in cerebellar development and how the abnormalities that occur due to RORα deficiency could explain NDD symptoms. We then focus on how RORα is linked to NDDs, particularly ASD and schizophrenia, and how its diverse extra-cerebral actions can explain the systemic components of these diseases. Finally, we discuss how RORα-deficiency is likely a driving force for NDDs through its induction of cerebellar developmental defects, which in turn affect downstream targets, and its regulation of extracerebral systems, such as inflammation, circadian rhythms, and sexual dimorphism.
Figure 2. RORα regulates multiple genes and plays
extensive roles in cerebellar development. (A) Key stages of PC development which are
regulated by RORα. These are at all stages from embryonic development to adult
maintenance. . (B) A schema showing the central role of RORα in
multiple cellular processes, that are modified in NDDs. When RORα is reduced
(central red circle), its regulation of gene transcription is altered. Here we
include the known RORα target genes that are also involved in NDDs. The effects
in red illustrate the induced abnormalities according to the direction of
change: estrogen and PC development are reduced, circadian rhythms are
perturbed, but inflammation and ROS are increased.
Sound sensitivity in autism and Nav1.2
At this point today’s post does get complicated.
Researchers have learnt that the sodium ion channel Nav1.2
(expressed by the SCN2A gene) can play a key role in hypersensitivity to sound
in autism.
Lack of these ion channels in the cells that produce myelin produces
“faulty auditory circuits”, with too much sound sensitivity.
An impairment in myelin structure can trigger cascading
effects on neuronal excitability. Sound sensitivity is just one example.
There is a great deal of evidence that genes involved in myelination are miss-expressed in many models of autism. Imaging studies have shown variations in myelination.
Scn2a deletion disrupts oligodendroglia
function: Implication for myelination, neural circuitry, and auditory
hypersensitivity in ASD
Autism spectrum disorder (ASD) is characterized by a complex etiology, with genetic determinants significantly influencing its manifestation. Among these, the Scn2a gene emerges as a pivotal player, crucially involved in both glial and neuronal functionality. This study elucidates the underexplored roles of Scn2a in oligodendrocytes, and its subsequent impact on myelination and auditory neural processes. The results reveal a nuanced interplay between oligodendrocytes and axons, where Scn2a deletion causes alterations in the intricate process of myelination. This disruption, in turn, instigates changes in axonal properties and neuronal activities at the single cell level. Furthermore, oligodendrocyte-specific Scn2a deletion compromises the integrity of neural circuitry within auditory pathways, leading to auditory hypersensitivity—a common sensory abnormality observed in ASD. Through transcriptional profiling, we identified alterations in the expression of myelin-associated genes, highlighting the cellular consequences engendered by Scn2a deletion. In summary, the findings provide unprecedented insights into the pathway from Scn2a deletion in oligodendrocytes to sensory abnormalities in ASD, underscoring the integral role of Scn2a-mediated myelination in auditory responses. This research thereby provides novel insights into the intricate tapestry of genetic and cellular interactions inherent in ASD.
Therefore, our study underscores the region-specific relationship between myelin integrity and ion channel distribution in the developing brain. We emphasize that any disturbances in myelin structure can trigger cascading effects on neuronal excitability and synaptic function in the CNS, especially at nerve terminals in the auditory nervous system.
How are Nav1.2 channels, encoded by Scn2a, involved in OL maturation and myelination? One possible explanation is that the activation of Nav1.2 may be pivotal for triggering Cav channel activation, leading to a Ca2+ flux within OLs, which is involved in OL proliferation, migration, and differentiation. Specifically, Ca2+ signaling facilitated by R-type Cav in myelin sheaths at paranodal regions, might influence the growth of myelin sheaths. To activate high-voltage activated calcium channels such as L- and R-Type efficiently, the activation of Nav1.2 channels should be required for depolarizing OL membrane to around -30 mV. Consequently, the synergic interplay between Nav1.2 and Cav channels could amplify calcium signaling in OLs, initiating the differentiation and maturation processes.
Defects in myelination can create a
spectrum of auditory dysfunctions, including hypersensitivity. Our results
demonstrated how OL-Scn2a is involved in the relationship between myelin
defects, neuronal excitability, and auditory pathology in ASD, potentially
paving the way for targeted therapeutic interventions.
One subject
that some people write to me repeatedly about is self-injurious behavior, so I
took note of the paper below.
Dopamine Transporter Binding Abnormalities Are Associated with Self-injurious Behavior in Autism Spectrum Disorder
Utilizing
single-photon emission computed tomography dopamine transporter scans (DaTscan)
we examined whether imaging markers of the dopaminergic system are related to
repetitive behaviors as assessed by the Repetitive Behavior Scale-Revised in
ASD.
Background:
Autism
spectrum disorder (ASD) is characterized by impairments in social
communication, and restricted repetitive behaviors. Self-injurious behaviors
are often observed in individuals with ASD. Dopamine is critical in reward,
memory, and motor control. Some propose the nigrostriatal motor pathway may be
altered in ASD, and alterations in dopamine are reported in some rodent models
based on specific ASD genes. Additionally, repetitive behaviors may to be
related to reward systems. Therefore, we examined the dopaminergic system,
using DaTscans, to explore its relationship with measures of repetitive
behavior in a clinical ASD population.
Design/Methods:
Twelve
participants (aged 18–27) with ASD were recruited from the Thompson Center for
Autism and Neurodevelopment and completed the Repetitive Behaviors Scale -
Revised (RBS-R). Of the 12 participants, 10 underwent a 45-minute DaTscan.
ANOVA was used to compare the dopamine imaging findings with the overall total
RB scores on the RBS-R. while other domains of the RBS-R were also investigated
in an exploratory manner.
Results:
Five of the participants had regional
deficits in dopamine transporter binding in the striatum on DaTscan.
Individuals with deficits on the DaTscan had significantly higher
Self-Injurious Endorsed Scores than those with normal scans.
Conclusions:
Half of the DaTscans obtained were
determined abnormal, and abnormal scans were associated with greater endorsing
of self-injurious behavior. Larger
samples are needed to confirm this, and determine the impact of laterality of
abnormalities, but this
preliminary work suggests a potential role the dopaminergic system in
self-injurious RBs. Elucidation of this relationship may be important
for future interventional outcomes, with potential impact on targeted
treatment, as the only currently approved medications for ASD are atypical
neuroleptics.
Dopamine
transporter binding abnormalities refer to deviations from the normal levels of
dopamine transporter (DAT) in the brain. DAT is a protein on the surface of
cells that reabsorbs dopamine from the synapse, regulating its availability.
Imaging
techniques like DAT scans (dopamine transporter scans) are used to assess DAT
levels. These scans measure the binding of radiotracers to DAT, with lower
binding indicating reduced DAT levels.
Dopamine
transporter binding abnormalities have been linked to various neurological and
psychiatric conditions, including:
• Parkinson's
disease: Degeneration of dopamine-producing neurons in the substantia nigra, a
hallmark of Parkinson's disease, leads to a significant decrease in dopamine
levels and DAT binding in the striatum.
• Attention
deficit hyperactivity disorder (ADHD): Some studies suggest that individuals
with ADHD may have abnormal DAT function, though the nature of the abnormality
(increased or decreased DAT) is debated.
• Autism
spectrum disorder (ASD): Research suggests that a subgroup of individuals with
ASD may have DAT abnormalities, potentially linked to repetitive behaviors and
social difficulties.
• Addiction:
Dopamine plays a central role in reward and motivation. Drugs like cocaine and
methamphetamine can cause long-term changes in DAT function, potentially
contributing to addiction.
DAT binding
abnormalities may not always translate to functional impairments.
Treatment
options for DAT binding abnormalities
Unfortunately,
medications that directly target Dopamine Transporter (DAT) binding
abnormalities do not exist.
In Parkinson's
disease the goal is to increase dopamine levels in the brain. Medications like
levodopa, a dopamine precursor, or dopamine agonists (drugs that mimic
dopamine) are used.
Conclusion
It certainly
is not easy to figure out how to treat autism and its troubling symptoms like
self-injury. Our reader currently trying to make sure his second child does not
have severe autism is wise to invest his time now.
Today we
added agmatine and choline to our list of preventative strategies to consider.
As regards strategies
to treat autism in children and adults, we see that the research very often is
repeating what has already been published over the past two decades.
Ion channels
do seem to be central to understanding and treating autism.