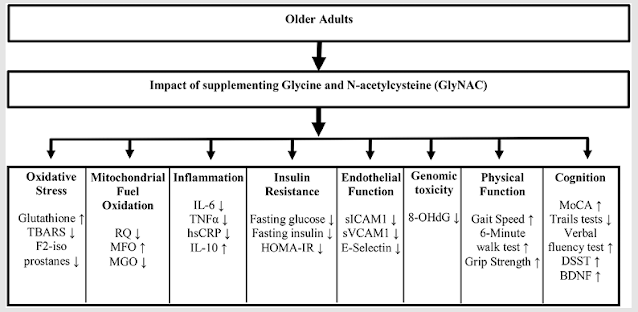
Diet can affect behaviour - but in some people much more so than others.
As the threshold for a diagnosis of autism, and indeed ADHD, is reduced more and more people may find a dietary solution.
Even people with a more severe neurological disorder may find dietary modification provides a benefit.
The big mistake is to think that all you can/need to do to treat disorders like autism is modify diet and pop a few vitamins and supplements.
In today’s post I grouped together causes, or contributors, to autism that fall in my “dietary” category.
- Food allergy
- Food intolerance
- Gluten related disorders
- Histamine intolerance
- Pesticides
- SIBO
- Propionic and Butyric acid (SCFAs)
- Probiotics and Fiber
- Low Glycemic Diet
- Ketogenic Diet
- FMT/MTT
- Fragile Gut
- Pancreatic Insufficiency
I do not dwell too much on diet in this blog probably because I have one son who has always had a near perfect diet, has no GI issues, but has autism and yet my other son, with an arguably poor self-restricted diet, is healthy, again with no GI issues and is totally neurotypical. Yes, perhaps if the son with autism had the restricted diet, he would have fared worse – we will never know.
Many people with severe autism have restricted diets, somewhat reinforced by their parents wanting to keep the peace.
People with autism are prone to auto-immune conditions and that will include food allergies (wheat, milk etc). Restricted diets lead to a microbiome that is equally restricted. Eating large amounts of pro-inflammatory junk food (chicken nuggets, french fries, sugar (sucrose and fructose), refined carbohydrate, processed meat etc) is a poor choice for anyone, autistic or not.
Improving diet does hold potential for many with autism and/or ADHD.
You have to choose for yourself what is relevant and what is a distraction in your N=1 case.
Food intolerance vs Food Allergy
Food intolerance refers to difficulty in digesting certain foods. It is important to note that food intolerance is different from food allergy.
Food allergies trigger the immune system, while food intolerance does not. Some people suffer digestive problems after eating certain foods, even though their immune system has not reacted - there is no histamine response.
Foods most commonly associated with food intolerance include dairy products, grains that contain gluten, and foods that cause intestinal gas build-up, such as beans and cabbage.
Here are some key points about food intolerance.
· Symptoms of food intolerance tend to take longer to appear than symptoms of allergies
- The symptoms are varied and can include, migraine, cough, and stomach ache
- Some food intolerance is caused by the lack of a particular enzyme
According to James Li, M.D., Ph.D., when it is an allergy, even small amounts result in symptoms, as may be the case with peanuts. Whereas, with food intolerance, tiny amounts will usually have no effect.
The symptoms of food intolerance generally take longer to emerge, compared to food allergies.
Onset typically occurs several hours after ingesting the offending food or compound and may persist for several hours or days. In some cases, symptoms may take 48 hours to arrive.
Some people are intolerant to several groups of foods, making it harder for doctors to determine whether it might be a chronic illness or food intolerance. Identifying which foods are the culprits can take a long time.
Enzyme deficiencies are a common cause of food intolerance.
People who are lactose intolerant do not have enough lactase, an enzyme that breaks down milk sugar (lactose) into smaller molecules that the body can break down further and absorb through the intestine. If lactose remains in the digestive tract, it can cause spasm, stomach ache, bloating, diarrhea, and gas.
People with an allergy to milk protein have similar symptoms to those with lactose intolerance; that is why lactose intolerant individuals are commonly misdiagnosed as allergic.
Histamine Intolerance
Many foods and drinks contain histamine.
Usually, the enzyme DAO, and to a lesser extent HNMT, breaks down ingested histamine, preventing it from being absorbed in the gut and entering the bloodstream. Within the brain histamine acts as a neurotransmitter.
Some factors can interfere with how DAO and HMNT work, or how much of these enzymes are present in the gut.
The common food additive Sodium Benzoate (E211) is a DAO-inhibitor. It is widely used in carbonated drinks, jams, fruit juice, pickles and condiments. Someone who is histamine intolerant will need to learn to avoid such foods.
Other common factors that interfere with DAO and HMNT levels include many prescription drugs, for example:
- airway medications, such as theophylline
- heart medications
- antibiotics
- antidepressants
- antipsychotics
- diuretics
- muscle relaxants
- pain medications
- gastrointestinal medicines
- nausea and gastroesophageal reflux disease, GERD
- malaria drugs
- tuberculosis medications
Pesticides
I think it is hardly surprising that pesticides can affect a developing brain, just like the lead that used to be added to petrol/gasoline.
The key time to avoid pesticides and toxic chemical is during pregnancy and early childhood.
Once you have got autism with MR/ID, it is probably rather late to worry about a small risk from a tiny exposure to pesticides from supermarket fruit and vegetables.
Exposure to common agricultural pesticides before birth and in the first year of life is associated with a small to moderately increased risk of autism spectrum disorder (ASD) compared with infants of women without such exposure, finds a study published in The BMJ today.
The researchers say their findings support efforts to prevent exposure to pesticides during pregnancy to protect a child's developing brain.
Experimental studies have suggested that common pesticides can affect normal brain development, and environmental exposures during early brain development are suspected to increase risk for autism spectrum disorders in children.
But studies examining pesticide exposure in the real world and risk of ASD are rare.
So, researchers at the University of California used registry records to identify 2,961 patients with a diagnosis of ASD -- including 445 with ASD with accompanying intellectual disability -- and 35,370 healthy ("control") patients of the same birth year and sex.
Participants were born between 1998 and 2010 in California's Central Valley, a heavily agricultural region, and 80% of cases were male.
Data from the California state-mandated Pesticide Use Registry were then integrated into a geographic information system tool to assess prenatal (before birth) and infant exposures to 11 commonly used pesticides (measured as pounds of pesticides applied per acre/month within 2 km of their mother's residence during pregnancy and exposure during developmental periods defined as yes vs no).
These pesticides were selected because of their high use and evidence indicating toxic effects on brain development.
After adjusting for potentially influential factors, the researchers found modest increases in ASD risk among offspring exposed to several pesticides (including glyphosate, chlorpyrifos, diazinon, malathion, permethrin, bifenthrin and methyl-bromide) before birth and during the first year of life, compared with controls.
Associations were strongest in those with ASD and intellectual disability, which represents the more severe end of the autism spectrum.
This is an observational study, and as such, can't establish cause, and the researchers point to some limitations, such as relying on patient records for details about diagnoses, and being unable to examine clinical outcomes.
Nevertheless, they say their study is by far the largest investigating pesticides and autism spectrum disorder to date and their findings back up earlier work in this field.
"Our findings suggest that ASD risk may increase with prenatal and infant exposure to several common ambient pesticides that impacted neurodevelopment in experimental studies," they write.
They call for further research to explore underlying mechanisms in the development of autism. However, from a public health and preventive medicine perspective, they say their findings "support the need to avoid prenatal and infant exposure to pesticides to protect the developing child's brain."
Gluten Free?
One long-known feature of autism is the loss of Purkinje cells, these cells are involved in motor skills and this probably contributes to clumsiness and poor handwriting in many people with autism. For good motor skills you need plenty of Purkinje cells, with plenty of myelin coating their axons.
An extreme cause of Purkinje cell loss in some people is a reaction to gluten, mainly in those with Celiac Disease (CD). The process is not fully understood but results in antibodies selectively destroying Purkinje cells and leading to a condition called Cerebellar Ataxia.
People sensitive to gluten, but not having Celiac disease, may also experience some ataxia as well as a wide range of auto-immune disorders that can include psychiatric manifestations.
I think a small number of people with autism do have non celiac gluten sensitivity (NCGS). Those people should feel better on a gluten free diet. A small number of people with severe autism may have undiagnosed Celiac disease.
Gluten related disorders
If you have one of the above conditions then avoid gluten.
If you do not have one of the above conditions, you are in the great majority and there is no point spending extra money to avoid gluten.
There is no reliable data, but an estimate is that 10-15% of people have some kind of gluten related disorder.
It is not surprising that a minority of people with autism respond to a gluten free diet, but the majority do not. Your N=1 case of autism could fall in either camp.
Source:- Dr Schär AG
Cerebellar ataxia (CA) is one of the most frequent neurological manifestations related to celiac disease (CD) (1), and may be the only and initial clinical manifestation of this disease (2) without any association with gastro-intestinal symptoms or malabsorption signs.
Gluten ataxia is purely cerebellar and involves the entire cerebellum (9). The clinical signs of CA are gait ataxia, limb ataxia, dysarthria, pyramidal signs, altered eyes motions, and progressive impairment of stability and erect position
The prolonged gluten consumption in patients with gluten ataxia leads to a progressive loss of Purkinje cells in the cerebellum. Patients with celiac disease and CA have a blood deficit of vitamin E
Non celiac gluten sensitivity (NCGS) is defined by clinical evidence of improvement of symptoms, following the introduction of GFD in the absence of enteropathy (22). Autoantibodies, such as TG2, are absent in NCGS. The presence of Anti Gliadin Antibodies (AGA) and particularly IgG AGA may be an indicator of NCGS in more than 50% of patients that refer to the gastroenterologist (23). Hadjivassiliou et al. (15) reported on 114 patients with NCGS and gluten ataxia (GA), 68 of which had circulating TG6 antibodies.
An early diagnosis of CA and gluten related disorders (GRD) increases the possibility to improve the neurological process (8); the clinical improvement is usually seen 1 year after starting the GFD (9) and continues for a period of about 2 years.
Cerebellar Ataxia is equally responsive to GFD in CD and NCGS patients.
Non celiac gluten sensitivity (NCGS) is a syndrome characterized by a cohort of symptoms related to the ingestion of gluten-containing food in subjects who are not affected by celiac disease (CD) or wheat allergy. The possibility of systemic manifestations in this condition has been suggested by some reports. In most cases they are characterized by vague symptoms such as ‘foggy mind’, headache, fatigue, joint and muscle pain, leg or arm numbness even if more specific complaints have been described. NCGS has an immune-related background. Indeed there is a strong evidence that a selective activation of innate immunity may be the trigger for NCGS inflammatory response. The most commonly autoimmune disorders associated to NCGS are Hashimoto thyroiditis, dermatitis herpetiformis, psoriasis and rheumatologic diseases. The predominance of Hashimoto thyroiditis represents an interesting finding, since it has been indirectly confirmed by an Italian study, showing that autoimmune thyroid disease is a risk factor for the evolution towards NCGS in a group of patients with minimal duodenal inflammation. On these bases, an autoimmune stigma in NCGS is strongly supported; it could be a characteristic feature that could help the diagnosis and be simultaneously managed. A possible neurological involvement has been underlined by NCGS association with gluten ataxia, gluten neuropathy and gluten encephalopathy. NCGS patients may show even psychiatric diseases such as depression, anxiety and psychosis. Finally, a link with functional disorders (irritable bowel syndrome and fibromyalgia) is a topic under discussion. In conclusion, the novelty of this matter has generated an expansion of literature data with the unavoidable consequence that some reports are often based on low levels of evidence. Therefore, only studies performed on large samples with the inclusion of control groups will be able to clearly establish whether the large information from the literature regarding extra-intestinal NCGS manifestations could be supported by evidence-based agreements.
SIBO
Small intestinal bacterial overgrowth (SIBO) is a serious condition affecting the small intestine. It occurs when bacteria that normally grow in other parts of the gut start growing in the small intestine. That causes pain and diarrhea. It can also lead to malnutrition as the bacteria start to use up the body’s nutrients.
If you have severe autism and live in rural China the study below suggests you have a 50:50 chance of having SIBO.
SIBO is measurable and treatable using mainstream medicine.
Don’t treat SIBO, if you do not have SIBO.
The aim of this study is to assess the prevalence of small intestinal bacterial overgrowth (SIBO) by hydrogen breath test in patients with autism spectrum disorders (ASD) with respect to a consistent control group. From 2011 to 2013, 310 children with ASD and 1240 sex- and age-matched typical children were enrolled in this study to undergo glucose breath test. The study participants were considered to exhibit SIBO when an increase in H2 of ≥20 ppm or CH4 of ≥10 ppm with respect to the fasting value was observed up to 60 min after the ingestion of glucose. Ninety-six children with autism suffered from SIBO, giving a prevalence rate of SIBO was 31.0% (95% CI 25.8-36.1%). In contrast, 9.3% of the typical children acknowledged SIBO. The difference between groups was statistically significant (P < 0.0001). The median Autism Treatment Evaluation Checklist (ATEC) score in the children with autism and with SIBO was significantly high when compared with the children without autism and without SIBO [98 (IQR, 45-120) vs. 63 (32-94), P < 0.001]. For the autism group, the 6-GI Severity Index (6-GSI) score was found to be strongly and significantly correlated with the total ATEC score (r = 0.639, P < 0.0001). SIBO was significantly associated with worse symptoms of autism, demonstrating that children with SIBO may significantly contribute to symptoms of autism.

Diarrhea was the most common SIBO symptom (71.0% of ASD patients), followed by abdominal pain (37.1%), and abnormal feces (30.0%). Children with autism and with SIBO were more likely from the rural area.
Furthermore, we found that SIBO was associated with worse symptoms of autism. However, it is difcult to establish whether the changes seen play a causative role or are merely a consequence of the disease. Interestingly, the effectiveness of oral, non-absorbable antibiotics in temporarily reducing symptoms of autism [28] suggests that the relationship may be causal, that is, we hypothesize that SIBO may significantly contribute to symptoms of autism in some children. Several possible mechanisms can be inferred. First, propionate has severe neurological effects in rats and Clostridia species are propionate producers. Studies by MacFabe et al. [29] have demonstrated that injecting propionate directly into specific regions of rat brains in vivo can cause significant behavioral problems. Second, differences in the microbiota may also result in altered microbial metabolism of aromatic amino acids, with consequent changes in systemic metabolites (as reflected in urinary metabolite profiles), which could lead to neurological symptoms [30]. Third, the microbiota could also be involved in the disease etiology via interactions with the immune system [31]. Some of the possible mechanisms outlined above are more likely to involve changes within the overall balance of the whole microbial community, while others may be exerted by specific bacteria. Fourth, SIBO leads to steatorrhea, vitamin B12 absorptive impairment and also injury to the small intestinal microvilli which itself causes malabsorption [32]. Zhang et al. [33] suggested that decrease in brain vitamin B12 status across the lifespan that may reflect an adaptation to increasing antioxidant demand, while accelerated deficits due to GSH deficiency may contribute to neurodevelopmental and neuropsychiatric disorders. Finally, many pathogenic Gram-negative bacteria contain lipopolysaccharide (LPS) in their cell walls, which can cause damage in various tissues including the brain [3]. LPS-induced inflammation in the brain increases permeability of the blood–brain barrier and facilitates an accumulation of high levels of mercury in the cerebrum, which may aggravate ASD symptoms.
There are different levels and types of SIBO. These distinctions matter when determining the most appropriate treatment. Depending on the extent of your condition, treatment may vary.
· hydrogen-predominant SIBO: The primary treatment is the antibiotic rifaximin.
· methane-predominant SIBO: This type of SIBO is harder to treat, and it may take longer to respond to treatment. Use rifaximin plus neomycin for these cases.
· recurrent SIBO: formulations of antimicrobial herbs can be used to treat recurrences and as an alternative for initial treatment of hydrogen- or methane-predominant SIBO.
As part of treatment follow a FODMAP (low fermentable oligosaccharides, disaccharides, monosaccharides and polyols) diet.
People taking acid reducing drugs for reflux/GERD/GORD might note that PPI-induced dysbiosis is considered a type of SIBO. PPIs are proton pump inhibitors like Nexium that are now more popular than Histamine H2 blockers like Zantac/Ranitidine.
Interestingly apple cider vinegar (ACV) can counter PPI-induced dysbiosis. Your small intestines need some acid. Your body relies on Sodium bicarbonate released by the pancreas to maintain pH levels, but it can only reduce acidity, not increase it. I imagine a swig of anything acidic would likely have a similar effect, although ACV has non acid-related effects.
If it is SIBO, get a genuine diagnosis, treat it and avoid it reoccurring
Propionic and Butyric Acids and SCFAs (Short Chained fatty Acids)
SCFAs are produced when dietary fiber is fermented in the colon. Acetate, propionate, and butyrate are the three most common SCFAs.
Avoid dysbiosis in your colon
You need fiber and butyrate-producing bacteria for a healthy colon.
Epithelial metabolism shapes the colonic microbiota.
Left: During gut homeostasis, obligate anaerobic bacteria convert fiber into fermentation products (butyrate) to maintain the epithelium in a metabolic state characterized by high oxygen consumption. This metabolic polarization of differentiated colonocytes (C2) maintains epithelial hypoxia (<1 oxygen="" span="" style="background: yellow; mso-highlight: yellow;" to="">limit the amount of oxygen (O
2) diffusing into the gut lumen. Right: A metabolic reorientation of terminally differentiated colonocytes toward low oxygen consumption (C1) increases the concentration of respiratory electron acceptors (O2 and NO3–) emanating from the epithelial surface, thereby causing a shift in the microbial community from obligate to facultative anaerobic bacteria. The color scale at the bottom indicates O2 levels. SC, stem cell; TA, undifferentiated transit-amplifying cell; C2, terminally differentiated C2-skewed colonocyte; C1, terminally differentiated C1-skewed colonocyte; GC, goblet cell; NO, nitric oxide.
Propionic acid leads to PTEN↓ Inflammation↑ Gliosis↑ Mitochondrial dysfunction ↑
We saw in an earlier post that propionic acid is used to cause reversible autism in a mouse model. When propionic acid is infused directly into rodents' brains, it produces reversible behaviors (e.g., hyperactivity, dystonia, social impairment, perseveration) and brain changes (e.g., innate neuroinflammation, glutathione depletion). In the mouse model, you just feed them some NAC and they switch back to regular happy mice.
We now know the details of what is happening to those mice. To a lesser extent if you produce a lot of propionic acid in your human gut some may well make it to your brain and produce a similar effect.
Propionic Acidemia (PA) is an inborn error of metabolism caused by mutations in propionyl‐CoA carboxylase (PCC). The disease is characterized by the systemic accumulation of propionic acid (PPA) and its toxic derivatives. Children with PA are at high risk of developing low muscle tone, cardiomyopathy, neutropenia, pancreatitis, and ultimately death.PA also has neurological manifestations that include a high risk of seizures and stroke, and a wide range of chronic psychological and cognitive sequelae, with intellectual disability and language impairments being the most frequently reported.
We know that in autism increasing PTEN is generally a good thing. Gliosis includes things like activating the brains immune cells (microglia) which we know is bad.
PPA is believed to cause systematic mitochondrial dysfunction (MD), as evidenced by increased free acyl-carnitine (cofactor used to transport long-chain and very-long-chain fatty-acids into the mitochondria) in rats exposed to PPA16.
PPA promotes gliosis and Pro-Inflammatory cytokines release
PTEN was reported to regulate radial glia cell proliferation in the early stages of neural development through inhibition of Akt pro-survival pathway26. Recent studies reported that PTEN is downregulated in autistic glial cells26,27, however, what triggers PTEN inhibition in ASD remains uncertain. In this study, data suggest that PPA binding to its receptor may lead to GPR41-induced PTEN inhibition, thereof allowing Akt survival pathway to proceed. As we demonstrated in Fig. 5, PPA seems to tamper with both PTEN and activated p-Akt levels. PTEN expression decreased with increased PPA concentration and vice versa for p-Akt. Noteworthy, PPA interfered with the amount of activated p-Akt but not Akt expression. This result further validates that PPA has no direct effect on Akt expression but rather downregulates PTEN expression. Consequently, this allows p-Akt to remain active which results in over-proliferation of glia-committed neural progenitor cells.
There are many studies on this subject and the one below is from December 2019.
Background: Autism spectrum disorders (ASD) prevalence is increasing, but its etiology remains elusive and its satisfactory effective treatment is not available. The microbiota-gut-brain axis can contribute to ASD pathology and may supply an effective and promising way for ameliorating the ASD symptoms. Herein, we explore the differences of the gut microbiota profiles, fecal short-chain fatty acids (SCFAs) and peripheral neurotransmitters between ASD children and typical development children, and whether the probiotics + fructo-oligosaccharide (FOS) intervention alters the gut microbiota profiles, SCFAs and neurotransmitters in ASD children.
Methods: This study was divided into two stages. At the discovery stage, we compared the difference of the gut microbiota profiles (using 16S rRNA sequencing), faecal SCFAs (using GC-MS) and plasma neurotransmitters (using UHPLC-MS/MS) between 26 ASD children and 24 TD children. Then, all the 26 ASD children participated into the intervention stage, and we measured the gut microbiota profiles, SCFAs and neurotransmitters at before and after probiotics + FOS (n = 16) or placebo supplementation (n = 10) for ASD children.
Findings: Firstly, the gut microbiota were in a state of dysbiosis and significantly lower levels of Bifidobacteriales and Bifidobacterium longum in ASD subjects found at the discovery stage. Compared with TD children, the significantly lower levels of acetic acid, propionic acid, and butyric acid, and a hyperserotonergic state (the increased 5-HT) and dopamine metabolism disorder (the decreased homovanillic acid) were observed in ASD children. Secondly, the increasing growth of beneficial bacteria (Bifidobacteriales and B. longum) and suppressing the growth of suspected pathogenic bacteria (Clostridium) emerged after the probiotics + FOS intervention, with significant improvements in the severity of autism (assessed by the ATEC), and gastrointestinal symptoms (assessed by the 6-GSI). With probiotics + FOS intervention, the above SCFAs in children with autism significantly elevated and approached to that of the typical development children. However, the levels of concentrations in fecal isobutyric acid and caproic acid after probiotics + FOS intervention were markedly higher than TD children, and the plasma zonulin downregulation as an intestinal permeability marker. Interestingly, our data demonstrated that the decreased 5-HT and 5-hydroxyindolacetic acid, as well as the increased kynurenine and homovanillic acid emerged after probiotics + FOS intervention. Our analysis of Spearman's rank correlation showed that Clostridium were significantly positive associated with 5-HT. However, the above-mentioned changes did not show in the placebo group for ASD children.
Interpretation: Our data suggest that gut microbiome is closely correlated with SCFAs and some neurotransmitters. The probiotics + FOS intervention can modulate the gut microbiome, SCFAs and some neurotransmitters in association with improved ASD symptoms, including a hyper-serotonergic state and dopamine metabolism disorder.
Medical Food (Probiotics) from one end and FMT from the other
There is some very clever research that will lead to medical food, containing specific bacteria, as a means of promoting specific chemical reactions in your gut, which then effect other parts of your body.
In earlier posts we saw how you could mimic the effect of the ketogenic diet in reducing seizures by using medical food.
We saw how one kind of childhood leukaemia can be prevented by taking a particular bacteria in medical food or yoghurt.
But Prof Greaves adds: "The most important implication is that most cases of childhood leukaemia are likely to be preventable."
His vision is giving children a safe cocktail of bacteria - such as in a yoghurt drink - that will help train their immune system.
Even some expensive drugs have been found to be effective only in the presence of specific gut bacteria. So, alongside the drug give that bacteria?
Of 30 drugs examined in vitro, the efficacy of 10 was found to be significantly inhibited by certain bacteria, while the same bacteria improved the efficacy of six others
Conceivably, there is potential for direct interaction between systemically administered drugs at various body sites in the course of infection or in the case of orally administered drugs and microbiota of the small intestine.
In conclusion, our data bring attention to the fact that internal bacteria can interact with a drug therapy and could under certain circumstances influence treatment efficacy and/or side effects.
The microbiota comes from the mother and ideally involves natural delivery and mother’s milk.
Early use and overuse of antibiotics will disrupt the microbiota.
The body’s immune system is calibrated very early in life, if mis-calibrated it will over/under-react for the rest of your life. Early exposure to bacteria is part of the calibration process, this is why having a pet indoors during pregnancy reduces the chance of a child having allergies. Also, good to be exposed to the other animals’ humans have evolved alongside (domesticated farmyard animals).
This takes us back to the idea of the Halogenone/Holobiont.
This can be summed up as give your body the bugs it evolved to expect, or don’t be surprised when things start going wrong (aberrant immune responses etc).
FMT/MTT
Another way of colonizing your gut with beneficial bacteria is repopulate it with someone else’s, so called microbiota transfer therapy (MTT), better known as fecal microbiota therapy (FMT) or more simply a poop transplant (PT, I suppose).
Trials show that taking probiotics orally often has little impact on what is growing in your gut. The effect is often short term and the new bacteria do not colonize their new host.
The idea of taking someone else’s feces/poo and inserting into a child with GI problems and autism may not sound very high tech, but in small trials it has shown to be beneficial.
Clearly there is potential to transfer things that might not be beneficial.
In the end I think someone will develop a synthesized lab-made product containing the many billions of “good” bacteria. Ultimately this could be a personalized medical product, tailored to the individual needs of the patient.
In a new study, researchers demonstrate long-term beneficial effects for children diagnosed with ASD through a revolutionary fecal transplant technique known as microbiota transfer therapy (MTT).
"It is very unusual to see steady gradual improvement after the conclusion of any treatment," said Adams. "We only conducted the long-term follow-up study after several families told us that their child was continuing to improve significantly." Krajmalnik-Brown stated that the data suggests that the MTT intervention transformed the gut environment into a healthier status, leading to long-term benefit on both GI and ASD symptoms.
Does this only work in people with autism who have GI symptoms? I would suspect it does, but would like to see some evidence.
Low Glycemic Diet
Hypoglycemia is low blood sugar that can cause headaches, weakness, and anxiety. Hyperglycemia refers to high levels of sugar, or glucose, in the blood.
Low blood sugar (hypoglycemia) can also mimic the symptoms of ADHD. Hypoglycemia in children may cause uncharacteristic aggression, hyperactivity, the inability to sit still, and the inability to concentrate.
High blood sugar (hyperglycemia) also negatively affects behavior.
Eating foods with a low Glycemic Index (GI) avoids spikes in blood sugar. Your body responds to blood glucose spikes by producing more insulin. This kind of diet is used by people with diabetes, but is actually good for everyone.
Anecdotal evidence from comments in this blog and elsewhere does give some support for this diet in humans. We also now have some scientific research that also looks inside the brain.
The number of people diagnosed with autism - a spectrum of disorders characterized by social avoidance, repetitive behaviors and difficulty communicating - has risen dramatically over the past two decades for reasons that are unclear. A diet recommended for diabetics ameliorated signs of autism in mice, researchers have found. Although preliminary and not yet tested in humans, the findings might offer clues to understanding one potential cause of autism.
Intriguingly, in the new study, the brains of mice modeling autism that were fed the high-glycemic index diet had drastically less doublecortin, a protein indicator of newly developing neurons, compared to predisposed mice on the low-glycemic index diet. The deficiency was especially obvious in a part of the brain that controls memory.
In addition, the brains of the high-glycemic index diet mice appeared to have greater numbers of activated microglia, the resident immune cells of the brain. Their brains also expressed more genes associated with inflammation, compared to the mice fed the low-glycemic index diet.
Other studies of human mothers and their children with autism have implicated the activation of the immune system. For the most part, these studies have focused on infection, which causes a bout of inflammation -- as opposed to a high-glycemic index diet, which causes chronic, low-level inflammation, Maher says.
The new study found that the diet might directly influence the ecosystem of bacteria in the gut. More complex starches are broken down by bacteria that live in the lower part of the gut, the large intestine. The group saw some evidence of that in the blood, detecting metabolites that could only have come from the gut in larger amounts in the animals fed the high-glycemic index diet.
'We were really surprised when we found molecules in the blood that others had reported could only be generated by gut bacteria,' Maher says. 'There were big differences in some of these compounds between the two diets.'
The Ketogenic Diet
I think the Ketogenic Diet (KD) is the cleverest diet because it will genuinely help a small number of people, but for entirely different reasons.
We saw that in people with seizures, the beneficial effect does not come from ketones, it comes from the high fat diet changing the microbiome and causing different bacteria to thrive. The researchers at UCLA then showed how this effect final reaches the brain where it affects GABA and Glutamate levels, and so prevents epileptic seizures.
In some people with a problem transporting glucose across the blood brain barrier (as in Alzheimer’s) or a problem with converting that glucose into ATP in mitochondria (someone with mitochondrial disease) the ketone BHB becomes an alternative fuel for cells. These people would benefit from the ketones produced naturally in your body, when you follow the high fat Ketogenic Diet.
Pancreatic Insufficiency
Pancreatic insufficiency is actually one of the few things I did actually test for a few years ago.
It has been suggested that some people with autism cannot digest proteins properly and that this results in a lack of amino acids that are needs to produce neurotransmitters and build other new proteins.
This might occur if the pancreas was not doing its job of producing the required digestive enzymes. The medical term is pancreatic insufficiency.
Almost 10 years ago a special digestive cocktail called CM-AT was entering the “final stage of testing”. It is still being tested.
The developer of CM-AT initially suggested that a biomarker existed for responders. They wanted to use chymotrypsin as a measure of pancreatic function. This test is an old one and in children is best known for cystic fibrosis.
Many other conditions lead to pancreatic insufficiency. It can be caused by pancreatitis, other causes of insufficiency may include celiac disease, Crohn disease, Zollinger-Ellison syndrome and Shwachman-Diamond Syndrome (SDS). I guess any condition causing too much stomach acid would also lead to pancreatic insufficiency (that is what Zollinger-Ellison syndrome does). What about just “Fragile Gut”, which seems common in autism?
First evidence on Chymotrypsin:-
Background:
Emerging research has suggested that some children with ASD appear to be at high risk for gastrointestinal concerns. It has also been noted that many children with ASD have diets that are highly self- selective, e.g., having a preference for carbohydrates. Although it remains unclear what the basis for GI disruption is, it may be the case that some children with ASD have insufficient levels of digestive enzymes needed to process some food types, e.g., protein. If a child has this deficiency, they cannot optimally digest a class of food (e.g., protein), their food avoidance may be related to unpleasant sequelae associated with its ingestion (e.g., under-digested meat feeling like “lead shot” in the stomach). The enzyme chymotrypsin digests protein into its component amino acids. Amino acids, especially essential amino acids, play a crucial role in the production of neurotransmitters (e.g., dopamine and serotonin), are regulators of gene expression, and form the building blocks for new proteins.
Objectives:
The objectives of this study were: 1) determine the prevalence of abnormal levels of the enzyme fecal chymotrypsin (FCT) in children with autism, and 2) to determine whether FCT levels are associated with severity of autistic symptomatology.
Methods:
Participants were 323 children between the ages of 3 and 8 years (261 boys; mean age: 5.8 yrs.) who met DSM-IV criteria for Autistic Disorder, as screened by the Social Communication Questionnaire (SCQ) and confirmed by Autism Diagnostic Interview-Revised (ADI-R) and clinical interview. FCT levels were assessed using photometric assay of stool samples (performed by Quest Diagnostics); FCT levels ≤ 12.6 U/g are considered abnormally/pathologically low. Severity of autistic symptomatology was assessed using the total score of the Social Communication Questionnaire (SCQ) and the ADI-R subscale scores.
Results:
Of the 323 children, 198 (61.3%) had abnormally low/pathological levels of FCT activity (<12.6 U/g; mean FCT level=7.34), while 38.7% had normal levels (>12.6 U/g; mean FCT level=18.92). Comparison of FCT level and autism symptom level (i.e., ADI-R subscale scores, SCQ total score) in all participants revealed no statistically significant associations between FCT level and severity of autistic symptoms. This finding suggests that lower FCT levels in children with autism are not associated with more severe autistic symptomatology.
Conclusions:
The presence of low FCT levels in a large subset of children with autism suggests that chymotrypsin deficiency may be a key feature in some children with ASD. This enzymatic deficiency may place these at higher risk for a suboptimal supply of amino acids, which may in turn possibly undermine their ability to produce neurotransmitters, regulate gene expression, and synthesize new proteins. These findings may inspire further research into the role of the pancreas and amino acid deficiency in autism, and in a broader sense, into the physiology and biochemistry of a subset of children with autism. It also provides rationale for investigating chymotrypsin replacement therapy in children with autism who exhibit FCT deficiency.
Fragile Gut in Autism

Robust vs. Fragile Gut Function in Children with ASD. (A) Robust Gut: the healthy gut displays robust digestion of proteins and simple sugars by the small intestine brush border enzymes that make these nutrients absorbable. After digestion, very few intact nutrients remain and indigestible polysaccharides (fiber) remain. This fiber is consumed by saccharolytic bacteria, which line most of the large intestine, and produce beneficial byproducts (such as short chain fatty acids). Undigested proteins or amino acids are consumed by putrefactive bacteria, which are few in number, and produce potentially harmful putrefactive metabolites that are easily detoxified. The blood and lymphatics in the villi do not directly interact with the lumen of the small intestine, preventing the interaction of antigenic food molecules with the underlying immune tissue. (B) Fragile Gut: the fragile gut of children with autism displays reduced digestive capacity. The inflammation and deterioration of the gut lining may cause reduced expression and activity of brush border disaccharidases and peptidases and greater amounts of intact simple sugars and proteinaceous substrates and less fermentable fiber. This proteinaceous substrate is consumed by the more prevalent putrefactive bacteria producing greater amounts of putrefactive metabolites, such as ammonia, phenols, and sulfides. The blood and lymphatics in the villi are in contact with the lumen due to the excessive inflammation and the undigested proteins in the intestine are able to directly pass. This process allows for interaction of antigenic proteins with immune tissue leading to an aberrant immune response and subsequent autoantibody production
Treatment of Pancreatic Insufficiency
Pancreatic enzymes are widely used to treat pancreatic insufficiency. A common product is Creon.
Pancreatin is used for pancreatic enzyme replacement therapy - it contains varying amounts of protease (trypsin, chymotrypsin, elasase), lipase and amylase, which help with the digestion of protein, fat and starch respectively.
In the above study it was important that there was NO correlation between lower Chymotrypsin and more severe autism.
I suspect that the best treatment is to treat “Fragile Gut” and then perhaps chymotrypsin will go back to normal all by itself.
Conclusion
There is no single therapy for autism applicable to all. There can never be, because many hundreds, if not thousands, of dysfunctions can lead to an autism diagnosis. You need to treat the dysfunction, not the vague observational diagnosis.
You do not need to treat someone else's dysfunction, perhaps in vogue on Facebook, or the holistic therapist's pet dysfunction, you need to treat your specific person's actual dysfunction.
It is clear that some people, from all parts of the autism spectrum, can/do benefit from dietary intervention, but you have to match the dietary intervention to the underlying dysfunction (autism is not the dysfunction).
Buying organic chicken nuggets is unlikely to help anyone’s autism. Stopping eating fried food might indeed do some marginal good, in some cases.
My son loves fish soups and meat-based soups, made the old-fashioned way, making a stock by boiling up the bones. It makes very tasty soup, but it did not make autism go away. Some people apparently buy bone broth to "heal the gut". If you regularly eat non-farmed oily fish there is no need for expensive fish oil supplements.
Identifying any genuine food allergies, food intolerance and unusual chemical allergies/intolerances, like histamine, should reduce the occurrence of autism flare-ups/exacerbations. Not all commercial food allergy testing is reliable, even though it might be expensive, it is just a business. It is another case of buyer beware, like all exotic tests.
You will find anecdotes of wonderful improvement using dietary intervention, but you will struggle to find peer-reviewed clinical trials showing similar results. This just shows that only a sub-group, at most, are responders.
Only a dietary therapy that matches a genuine underlying dysfunction can be beneficial. The same is equally true for all drug interventions for autism.
If your only therapies are dietary, you have really only scratched the surface of ways to potentially treat autism. If you live in a country with no science-based autism MDs, you will have to do plenty of homework yourself.
For most people the best source of gut bacteria is likely a varied diet and this should be accompanied by a diet rich in fiber.
The many people with autism who have a very restricted diet and GI issues would seem the most likely to benefit from dietary interventions; but that might be stating the obvious. Can people with severe autism, but a varied diet and no GI problems benefit from dietary intervention? This is certainly possible and so should be considered, but looks less likely.
Commercially available probiotics range from quite potent ones that do have immunomodulatory effects, to mild ones to treat stomach upsets, to others that do very little. Some people respond negatively to specific probiotics that might be beneficial to another person.
Modifying the microbiome is the target of many new medical products in development. These products are not related to GI problems, they are using the microbiome to produce chemicals in the gut that then get absorbed and act just like a drug does. A slow sustained release of such chemicals inside the gut can be more beneficial that taking a drug by mouth, or nose or vein. Oxytocin produced in the gut is a good example.
I think dietary autism therapies require even more trial and error than pharmaceutical autism therapies. The bigger the effort and cost, the bigger the potential placebo effect becomes.
On the plus side, dietary therapy is easy to access, which is why it is so popular.
This post was not overly complicated, but it was long.