Source: Epiphany ASD Blog
Today’s post is very relevant to dementia, relevant to schizophrenia and diabetes and I believe some autism, including that of my son; agmatine is part of his Polypill therapy.
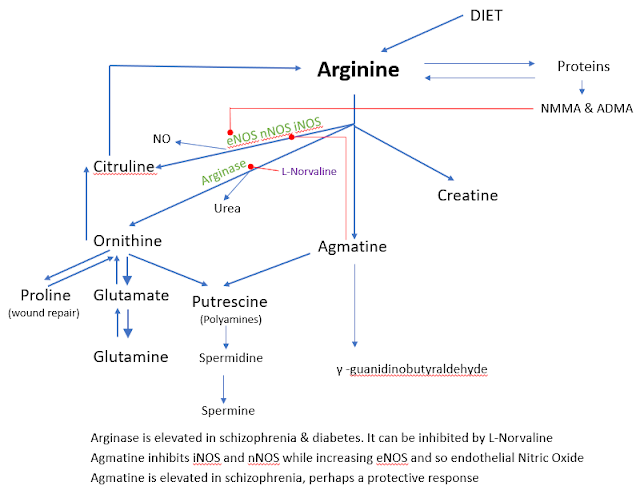
Arginine is highly versatile amino acid and you need the arginine metabolism to be working correctly, particularly in your brain.
Arginine is a widely available from diet and can be produced from citrulline and indirectly from glutamine; so you are unlikely to be deficient in arginine, except in your brain and particularly if you have Alzheimer’s.
In Alzheimer’s it has been shown that the microglia in effect destroy arginine in the brain and this may play a role in what initiates the disease.
Research has suggested that a deficiency in polyamines, another derivative of Arginine, is a feature of dementia.
A deficiency of arginine in the brain will likely cause a deficiency of polyamines.
Your body needs nitric oxide to maintain a healthy blood pressure and this requires arginine to follow the blue line in the above chart towards citrulline and be converted by eNOS. In most older people this does not happen and oxidative stress appears to be a big part of the problem.
Agmatine – good
Agmatine has been shown in research to have a benefit in Alzheimer’s.
This could be due to increased eNOS improving blood flow, an increase in Polyamines, or by reducing insulin resistance in the brain. Recall those studies of intranasal insulin? We had "type 3 diabetes", which was a brain-specific blunting of insulin.
https://www.ncbi.nlm.nih.gov/pubmed/27810390
"Agmatine administration rescued the reduction in insulin signalling, which in turn reduced the accumulation of Aβ and p-tau in the brain. Furthermore, agmatine treatment also reduced cognitive decline. Agmatine attenuated the occurrence of AD in T2DM mice via the activation of the blunted insulin signal"
Methylarginines – not good
Two by-products of arginine are bad for you in the way Agmatine is good for you.
Nitric Oxide is produced via iNOS, nNos and eNOS. In simple terms we want nitric oxide to be produced in the endothelium, the name for cells that line the interior surface of blood vessels and lymphatic vessels, To achieve this we needs lots of the enzyme eNOS and not much iNOS or nNOS, this is one of Agmatine’s jobs.
Two derivatives of arginine/proteins in the body with very long names are abbreviated to NMMA and ADMA. They both inhibit eNOS and so will restrict blood flow and this will appear as elevated blood pressure.
Endogenous methylarginines, N(G),N(G)-dimethyl-L-arginine (asymmetric dimethylarginine, ADMA), N(G)-N('G)-dimethyl-L-arginine (symmetric dimethylarginine; SDMA), and N(G)-monomethyl-L-arginine (monomethyl arginine; NMMA) are supposed to be produced in human body through the methylation of protein arginine residues by protein arginine methyltransferases (PRMT) and released during proteolysis of the methylated proteins. Micromolar concentration of ADMA and NMMA can compete with arginine for nitric oxide synthase (NOS) reducing nitric oxide (NO) formation, whereas SDMA does not. Indeed, increased ADMA and SDMA plasma levels or a decreased arginine/ADMA ratio is related with risk factors for chronic kidney disease and cardiovascular disease. To the best of our knowledge the exogenous presence of methylarginines, like that in fruits and vegetables, has never been described so far. Here, we report the finding that methylarginines are ubiquitous in vegetables which represent an important part of human daily diet. Some of these vegetables contain discrete amounts of ADMA, SDMA, and NMMA. Specifically, among the vegetables examined, soybean, rye, sweet pepper, broad bean, and potato contain the highest ADMA and NMMA mean levels. Our results establish that the three methylarginines, in addition to being produced endogenously, can also be taken daily through the diet in conspicuous amounts. We propose that the contribution of the methylarginines contained in the vegetables of daily diet should be taken into account when the association between vegetable assumption and their levels is evaluated in clinical studies. Furthermore, a comprehensive understanding on the role of the digestive breakdown process and intestinal absorption grade of the methylarginines contained in vegetables is now needed.
ADMA
Asymmetric dimethylarginine (ADMA) is a naturally occurring chemical found in blood plasma. It is closely related to L-arginine. ADMA interferes with L-arginine in the production of nitric oxide (NO), a key chemical involved in normal endothelial function and, by extension, cardiovascular health. ADMA inhibits eNOS, which in simple terms is the good NOS, the other two being iNOS and nNOS.
NMMA (NG-monomethyl-l-arginine, or just called Targinine)
The following study is very interesting for your older relatives. As we already know oxidative stress is a feature of aging. Many people have high blood pressure in old age. Nitric Oxide (NO) is needed keep blood vessels wide open. In old age (>60) oxidative stress reduces NO availability to nothing.
Since oxidative stress is reversible (in this study vitamin C was used) you wonder why more older people, particularly with high blood pressure, do not take entioxidants.
A novel finding of the present study is that in normotensive subjects, the reduction in endothelial function associated with aging seems to be mediated by a progressive reduction of NO availability, inasmuch as the inhibiting effect of L-NMMA on acetylcholine-induced vasodilation was progressively impaired by advancing age. It is worth noting that after the age of 60 years, the inhibiting effect of L-NMMA on response to acetylcholine was very weak, suggesting that in aged individuals NO availability is almost totally compromised. To assess the possible role exerted by oxidative stress, we tested the antioxidant vitamin C.19 Up to the age of 60 years, despite the evident decline in endothelium-dependent vasodilation, vitamin C did not modify the response to acetylcholine. In contrast, in the oldest individuals (age >60 years) characterized by a profound alteration in NO availability, vitamin C not only enhanced the response to the endothelial agonist but also restored the inhibiting effect of L-NMMA on vasodilation to acetylcholine. Thus, in the present study, the use of L-NMMA and vitamin C, never tested before in investigating the mechanisms responsible for the previously demonstrated age-related endothelial dysfunction in humans,17 seems to indicate that the progressive impairment in endothelium-dependent vasodilation is caused by a progressive alteration of the l-arginine-NO pathway. Only in old age (after ≈60 years) does the production of oxidative stress appear, leading to the complete compromise of NO availability.
Arginase
Arginase is an enzyme that acts as the catalyst for the reaction.
People with schizophrenia and also people with diabetes tend to have high levels of Arginase. This will affect how arginine is metabolized. If arginase is increased there is less arginine that can go towards creatine, citrulline or agmatine.
Going towards citrulline involves the production of nitric oxide NO. Now in schizophrenia we see a reduction in the good type of NO, that produced in the endothelium, the cells that line the interior surface of blood vessels and lymphatic vessels. As a result, we vascular dysfunction in schizophrenia.
Agmatine is also elevated in schizophrenia, which may be one of those feedback loops since agmatine will inhibit iNOS, nNOS while increasing eNOS
So where is there a reduction in Arginine in schizophrenia?
Well it looks like it is creatine which takes the hit.
“Patients with schizophrenia had a statistically significant reduction in Cr levels as compared with controls; bipolar disorder patients showed no difference in Cr as compared with controls”
In people with elevated arginase a useful strategy might be to use an arginase inhibitor.
The next paper highlights the arginase inhibitor I favour, which is L-norvaline. The paper is from Kursk university. Kursk gave its name to the nuclear-powered submarine that was lost in the Barents Sea in 2000 and triggered a new international cooperation to rescue stricken submarines. The Battle of Kursk was the largest tank battle of all time and the final major offensive by the Germans against the Russians in World War 2, where Hitler wanted to cut off a large bulge in the front line and trap a lot of Russians. Thanks to some clever English mathematicians, encrypted German communications were readable and the Russians repositioned their forces in advance, allowing them to counter attack. The Allies then invaded Sicily and that was the end for the Germans in Russia.
The present research shows expressed endothelium-protective property of arginase inhibitor, L-norvaline, characterized by decrease of coefficient of endothelial dysfunction and the approached its application to a group of intact animals. In other words, L-norvaline prevents the development of systemic endothelial dysfunctions in L-NAME- and methionine-induced NO deficiency.
Age-induced memory impairment (AMI)
Now we move to Polyamines that are on the bottom left my graphic at the start of this post. Spermidine and Spermine are very beneficial derivatives of arginine that most older people will be lacking. Autophagy is the cellular garbage disposal service that is dysfunction in many neurological disorders. We generally want more autophagy.
The aging process drives the progressive deterioration of an organism and is thus subject to a complex interplay of regulatory and executing mechanisms. Our understanding of this process eventually aims at the delay and/or prevention of age-related pathologies, among them the age-dependent decrease in cognitive performance (e.g., learning and memory). Using the fruit fly Drosophila melanogaster, which combines a generally high mechanistic conservation with an efficient experimental access regarding aging and memory studies, we have recently unveiled a protective function of polyamines (including spermidine) against age-induced memory impairment (AMI). The flies’ age-dependent decline of aversive olfactory memory, an established model for AMI, can be rescued by both pharmacological treatment with spermidine and genetic modulation that increases endogenous polyamine levels. Notably, we find that this effect strictly depends on autophagy, which is remarkable in light of the fact that autophagy is considered a key regulator of aging in other contexts. Given that polyamines in general and spermidine in particular are endogenous metabolites, our findings place them as candidate target substances for AMI treatment.
Aging is the most important risk factor for cardiovascular disease (CVD). Slowing or reversing the physiological impact of heart aging may reduce morbidity and mortality associated with age-related CVD. The polyamines, spermine (SP) and spermidine (SPD) are essential for cell growth, differentiation and apoptosis, and levels of both decline with age. To explore the effects of these polyamines on heart aging, we administered SP or SPD intraperitoneally to 22- to 24-month-old rats for 6 weeks. Both treatments reversed and inhibited age-related myocardial morphology alterations, myocardial fibrosis, and cell apoptosis. Using combined proteomics and metabolomics analyses, we identified proteins and metabolites up- or downregulated by SP and SPD in aging rat hearts. SP upregulated 51 proteins and 28 metabolites while downregulating 80 proteins and 29 metabolites. SPD upregulated 44 proteins and 24 metabolites and downregulated 84 proteins and 176 metabolites. These molecules were mainly associated with immune responses, blood coagulation, lipid metabolism, and glutathione metabolism pathways. Our study provides novel molecular information on the cardioprotective effects of polyamines in the aging heart, and supports the notion that SP and SPD are potential clinical therapeutics targeting heart disease
Figure 1. summarizes the suggestion that spermidine-triggered restoration of autophagy protects synapses from age-induced changes, and thus delays the normally occurring decline of memory formation. Given that spermidine is a physiologic, easy administrable substance, future research may consider its supplementation to counter age-dependent dementia.
Spermidine operates directly at presynaptic active zone scaffolds (composed of Brp/bruchpilot protein) to allow for an autophagy-dependent homeostatic regulation of these specializations. In effect, spermidine protects learning efficacy from aging-induced decline.
Having your longevity and eating too
Although caloric restriction has clear benefits for maximizing health span and life span, it is sufficiently unpleasant that few humans stick to it. Madeo et al. review evidence that increased intake of the polyamine spermidine appears to reproduce many of the healthful effects of caloric restriction, and they explain its cellular actions, which include enhancement of autophagy and protein deacetylation. Spermidine is found in foods such as wheat germ, soybeans, nuts, and some fruits and vegetables and produced by the microbiota. Increased uptake of spermidine has protective effects against cancer, metabolic disease, heart disease, and neurodegeneration.
Although spermidine induces autophagy and autophagy inhibition curtails many of the health-promoting effects of spermidine, additional mechanisms have been proposed to explain the beneficial effects of spermidine on aging. These potentially autophagy-independent mechanisms include direct antioxidant and metabolic effects on arginine bioavailability and nitric oxide (NO) production. However, it has not been formally determined whether these routes act in a completely autophagy-independent manner or are interrelated with autophagy (in an additive or synergistic way) (see the figure), and it will be important to define actionable molecular targets that explain the beneficial effects of spermidine in diverse pathophysiological settings. In this sense, it will also be of interest to explore synergisms of spermidine with other CRMs that initially act through different mechanisms.


It is a surprise that those long-lived Japanese eat Natto? Also, it is a good source of vitamin K2 and importantly it is an estrogen and so an ERβ agonist.
Not all probiotics are helpful to produce polyamines and one well known probiotic, VSL#3, has been shown to reduce their level. Choose your bacteria very carefully.
Here the probiotic strain Bifidobacterium animalis subsp. lactis LKM512 is used to increase polyamine production
Alzheimer’s and Arginine
In a fairly recent study it was suggested that the immune system in the brain is being suppressed and the microglia are slightly mutated along with the over-expression of arginase. Arginase is the enzyme that coverts arginine to ornithine plus urea.
So, in Alzheimer’s there will be a lack of arginine available for its other purposes.
So, we would expect a lack of creatine, agmatine and citrulline. Along the way we should see less Nitric Oxide.
Based on my graphic above, it would seem that L-Norvaline should improve the outcome in Alzheimer’s mice.
We already know that Agmatine improves Alzheimer’s mice, as we now should expect.
So, my cocktail for an aging mouse would be: -
· L-Norvaline (used by body builders)
· Agmatine (used by body builders)
· Creatine (used by body builders)
· Natto/wheatgerm/ LKM512 probiotic
· Vitamin C or NAC
· Citrulline (used by body builders)
· Betanin (an approved food colour additive, see below)
Served with cheese, naturally.
Alzheimer’s study suggests immune cells chew up an important amino acid
Increasingly, evidence supports the idea that the immune system, which protects our bodies from foreign invaders, plays a part in Alzheimer’s disease. But the exact role of immunity in the disease is still a mystery. A new Duke University study in mice suggests that in Alzheimer’s disease, certain immune cells that normally protect the brain begin to abnormally consume an important nutrient: arginine. Blocking this process with a small-molecule drug prevented the characteristic brain plaques and memory loss in a mouse model of the disease. Published April 15 in the Journal of Neuroscience, the new research not only points to a new potential cause of Alzheimer’s but also may eventually lead to a new treatment strategy. “If indeed arginine consumption is so important to the disease process, maybe we could block it and reverse the disease,” said senior author Carol Colton, professor of neurology at the Duke University School of Medicine, and a member of the Duke Institute for Brain Sciences. The brains of people with Alzheimer’s disease show two hallmarks -- ‘plaques’ and ‘tangles’ -- that researchers have puzzled over for some time. Plaques are the build-up of sticky proteins called beta amyloid, and tangles are twisted strands of a protein called tau. In the study, the scientists used a type of mouse, called CVN-AD, that they had created several years ago by swapping out a handful of important genes to make the animal’s immune system more similar to a human’s. Compared with other mice used in Alzheimer’s research, the CVN-AD mouse has it all: plaques and tangles, behaviour changes, and neuron loss. In addition, the gradual onset of these symptoms in the CVN-AD mouse gave researchers a chance to study its brain over time and to focus on how the disease begins, said the study’s first author Matthew Kan, an MD/PhD student in Colton’s lab. Looking for immune abnormalities throughout the lifespan of the mice, the group found that most immune system components stayed the same in number, but a type of brain-resident immune cells called microglia that are known first responders to infection begin to divide and change early in the disease. The microglia express a molecule, CD11c, on their surface. Isolating these cells and analyzing their patterns of gene activity, the scientists found heightened expression of genes associated with suppression of the immune system. They also found dampened expression of genes that work to ramp up the immune system. “It’s surprising, because [suppression of the immune system is] not what the field has been thinking is happening in AD,” Kan said. Instead, scientists have previously assumed that the brain releases molecules involved in ramping up the immune system, that supposedly damage the brain. The group did find CD11c microglia and arginase, an enzyme that breaks down arginine, are highly expressed in regions of the brain involved in memory, in the same regions where neurons had died. Blocking arginase using the small drug difluoromethylornithine (DFMO) before the start of symptoms in the mice, the scientists saw fewer CD11c microglia and plaques develop in their brains. These mice performed better on memory tests. “All of this suggests to us that if you can block this local process of amino acid deprivation, then you can protect -- the mouse, at least -- from Alzheimer’s disease,” Kan said. DFMO is being investigated in human clinical trials to treat some types of cancer, but it hasn’t been tested as a potential therapy for Alzheimer’s. In the new study, Colton’s group administered it before the onset of symptoms; now they are investigating whether DFMO can treat features of Alzheimer’s after they appear. Does the study suggest that people should eat more arginine or take dietary supplements? The answer is ‘no,’ Colton said, partly because a dense mesh of cells and blood vessels called the blood-brain barrier determines how much arginine will enter the brain. Eating more arginine may not help more get into the sites of the brain that need it. Besides, if the scientists’ theory is correct, then the enzyme arginase, unless it’s blocked, would still break down the arginine. “We see this study opening the doors to thinking about Alzheimer’s in a completely different way, to break the stalemate of ideas in AD," Colton said. "The field has been driven by amyloid for the past 15, 20 years and we have to look at other things because we still do not understand the mechanism of disease or how to develop effective therapeutics
The full study: -
The pathogenesis of Alzheimer's disease (AD) is a critical unsolved question; and although recent studies have demonstrated a strong association between altered brain immune responses and disease progression, the mechanistic cause of neuronal dysfunction and death is unknown. We have previously described the unique CVN-AD mouse model of AD, in which immune-mediated nitric oxide is lowered to mimic human levels, resulting in a mouse model that demonstrates the cardinal features of AD, including amyloid deposition, hyperphosphorylated and aggregated tau, behavioral changes, and age-dependent hippocampal neuronal loss. Using this mouse model, we studied longitudinal changes in brain immunity in relation to neuronal loss and, contrary to the predominant view that AD pathology is driven by proinflammatory factors, we find that the pathology in CVN-AD mice is driven by local immune suppression. Areas of hippocampal neuronal death are associated with the presence of immunosuppressive CD11c(+) microglia and extracellular arginase, resulting in arginine catabolism and reduced levels of total brain arginine. Pharmacologic disruption of the arginine utilization pathway by an inhibitor of arginase and ornithine decarboxylase protected the mice from AD-like pathology and significantly decreased CD11c expression. Our findings strongly implicate local immune-mediated amino acid catabolism as a novel and potentially critical mechanism mediating the age-dependent and regional loss of neurons in humans with AD.
So Arginine for Alzheimer’s? Not so simple
Eating more arginine is not an effective way to increase the level of arginine in your brain and also the high level of arginase might just soak it all up anyway.
Other science does suggest that there are other ways to increase the amount of arginine in your brain, such as L-citrulline. We have already seen that we can inhibit arginase with L-norvaline among other things.
Betanin for Alzheimer’s
Since we are on Alzheimer’s, we might as well include another clever idea.
Our reader Tyler highlighted another interesting Alzheimer’s study, which suggests preventing/treating Alzheimer’s with Betanin, the pigment in beet root.
This might sound mad, but is deadly serious. The research showed that Betanin inhibits the formation of the trademark beta-amyloid plaques that define Alzheimer’s. No plaques, no Alzheimer’s.
Beetroot has already been featured in this blog; it has numerous health benefits.
To lower blood pressure and increase exercise endurance it is the nitrates that are helpful, but beetroot has numerous other effects; it even increases insulin sensitivity, so is a good choice for diabetics and pre-diabetics.
Betanin without the beetroot?
Betanin has such a strong colour it is used commercially as a food colourant, it appears as E162 on the label. In Europe it is called Beetroot red E162 and is inexpensive.
Personally, I take my betanin with the rest of the beetroot.
Vascular Dementia - before I forget
Vascular dementia is the easiest type of cognitive impairment to understand. Reduced blood flow to the brain, most likely due to reasons including a loss of endothelial nitric oxide, effectively starves the brain. We saw how cocoa flavanols improve blood flow and hence mild cognitive impairment, this is via an NO-dependent mechanism that nobody fully understands. In autism things get more complicated and we saw in earlier posts that we seem to have unstable blood flow rather than just reduced blood flow. Nonetheless, improving cerebral blood flow may well be useful for some people with autism; so more eNOS and not too much arginase, cocoa flavanols may well be beneficial. Antioxidants are hopefully already being taken.
Conclusion
I was surprised just how much in the post can be implemented today with no prescription medication.
It is no surprise that certain diets (Mediterranean/Okinawan) promote not only longevity but also an extended healthy life expectancy.
I think there are some tips here for fine tuning out of balance brains found in autism, schizophrenia and bipolar.
I hope someone trials my cocktail on an Alzheimer’s mouse and a regular older mouse.
· L-Norvaline and Citrulline
· Agmatine
· Creatine
· Natto/wheatgerm/ LKM512 probiotic
· Vitamin C or NAC
· Betanin
I suspect this cocktail would be more effective than Donepezil or Memantine, neither of which address the underlying cause of Alzheimer's disease. In reality some of the above might not even be needed (e.g. creatine and citrulline).
Agmatine as an alternative for some people who respond to intranasal insulin is an interesting idea. Research seems to have stalled because the preservative in the insulin causes irritation inside the nose.
Note: Creatine deficiency is a known cause of MR/ID/Autism and some types are treatable https://creatineinfo.org/. It is detectable by Magnetic Resonance Spectroscopy or by measuring creatine levels in plasma and urine. Babies born with creatine deficiency may exhibit hypotonia (floppy baby syndrome) due to weak muscles.